Clinical Significance of WISP-1 and VEGF-A Expression Levels in Elderly Patients with Esophageal Squamous Cell Carcinoma
-
摘要:目的
探索WNT1诱导信号通路蛋白-1(WISP-1)及血管内皮生长因子-A(VEGF-A)在老年食管鳞状细胞癌(ESCC)患者中作为新的分子标志物的临床价值。
方法将老年ESCC分为65~70岁组与>70岁组,分别应用免疫组织化学染色法与RT-PCR检测WISP-1、VEGF-A蛋白与mRNA的表达。Log rank检验分析临床病理特征与食管鳞癌预后的关系。
结果ESCC组织中WISP-1阳性表达率明显高于癌旁组织(53.33% vs. 35.42%,P=0.045)、>70岁及有淋巴结转移的患者显著高于65~70岁(P=0.047)及无淋巴结转移的患者(P=0.032)。ESCC组织中VEGF-A蛋白的阳性表达率显著高于癌旁组织(50.00% vs. 20.83%,P=0.001),浸润程度T3~T4期、有淋巴结转移、TNM分期Ⅲ~Ⅳ期显著高于T1~T2、无淋巴结转移、Ⅰ~Ⅱ期患者(P=0.030、0.006、0.010)。WISP-1 mRNA和VEGF-A mRNA表达水平明显高于癌旁组织(P<0.05)。单因素分析结果表明WISP-1及VEGF-A表达水平与老年ESCC的预后相关(P<0.05),多因素分析结果表明淋巴结转移是影响老年ESCC患者预后的独立危险因素。
结论WISP-1及VEGF-A与老年ESCC患者恶性行为相关,且年龄越大,WISP-1表达越高,有作为老年ESCC新的分子标志物的潜能。
Abstract:ObjectiveTo investigate the clinical value of WNT1 inducible signaling pathway protein 1 (WISP-1) and vascular endothelial growth factor-A (VEGF-A) as novel molecular markers of esophageal squamous cell carcinoma (ESCC) in elderly patients.
MethodsElderly patients with ESCC were divided into 65-70 years old group and >70 years old group. Immunohistochemical staining and RT-PCR were used to detect the expression levels of WISP-1 and VEGF-A proteins and mRNAs, respectively. Log rank test was used to analyze correlation between clinicopathological features and prognosis of ESCC in the elderly.
ResultsThe positive expression rate of WISP-1 in ESCC tissues was significantly higher than that in para-cancer tissues (53.33% vs. 35.42%, P=0.045). The positive expression rate of WISP-1 in patients over 70 years old and with lymph node metastasis was significantly higher than that in patients aged 65-70 years and without lymph node metastasis (P=0.047 and 0.032). The positive expression rate of VEGF-A protein in ESCC tissues was significantly higher than that in para-cancer tissues (50.00% vs. 20.83%, P=0.001). The positive expression rate of VEGF-A in patients with depth of infiltration T3-T4 stage, lymph node metastasis, and TNM Ⅲ-Ⅳ stage was significantly higher than that in patients with T1-T2 stage, no lymph node metastasis, and Ⅰ-Ⅱ stage (P=0.030, 0.006, and 0.010, respectively). The mRNA expression levels of these two molecules were significantly higher in ESCC tissues than in para-cancer tissues (P<0.05), and the results of univariate analysis showed that the expression levels of WISP-1 and VEGF-A were correlated with the prognosis of ESCC in the elderly (P<0.05). The results of multivariate analysis showed that lymph node metastasis was the independent risk factor affecting the prognosis of elderly patients with ESCC.
ConclusionWISP-1 and VEGF-A are associated with malignant behavior in elderly patients with ESCC. The expression of WISP-1 increased with age, and it has potential as a novel molecular marker of ESCC in elderly patients.
-
Key words:
- Elderly /
- Esophageal squamous cell carcinoma /
- WISP-1 /
- VEGF-A
-
0 引言
食管鳞状细胞癌(Esophageal squamous cell carcinoma, ESCC)是我国的主要致死性恶性肿瘤之一[1],5年生存率仅为30%左右,老年癌症患者(≥65岁)有着明显的生存劣势,大多数患者5年生存率都随着年龄的增长明显下降[2]。研究者一直努力寻找及验证更有临床应用价值的新的分子标志物以更精准地评估患者,延长生存期。Wnt1诱导信号通路蛋白-1(WNT1 inducible signaling pathway protein 1, WISP-1)与多种癌症的进展相关,血管内皮生长因子-A(Vascular endothelial growth factor A, VEGF-A)是临床抗血管生成药物针对的重要靶点。已发现上述两分子在骨肉瘤及胃癌研究中存在一定的相关性[3-4],WISP-1高表达提示ESCC患者预后较差[5]。但WISP-1及VEGF-A在老年ESCC患者中的表达、与临床病理特征的关系及二者相关性尚未见报道。实施积极应对人口老龄化是国家战略,因而本课题选择老年ESCC患者进行上述两分子研究,期望在老年ESCC患者中发现有临床意义的新的生物标志物。
1 资料与方法
1.1 研究对象
本研究以老年ESCC患者为研究对象,年龄分组参照国际类似研究,老年患者定义为年龄≥65岁的患者,再将其细分为65~70岁与>70岁组[6],肿瘤大小分为≤4 cm与>4 cm组[7]。TNM分期参照AJCC指南第八版[8]。
1.1.1 样本来源
蜡块组织:收集2015年9月至2018年12月在本院明确诊断为ESCC并行根治性切除术的90例老年患者临床资料及其ESCC组织蜡块标本,另收集48例配对的癌旁食管黏膜组织(距离癌组织5 cm以远取材)蜡块标本。
新鲜组织:收集2018年10月至2022年12月于本院明确诊断为ESCC并行根治性切除术的30例老年患者的癌组织标本及其配对癌旁食管黏膜组织(距离癌组织5 cm以远取材)标本。标本离体后立即在手术室切取所需标本,取材时避开肿瘤坏死区域,置于标本袋中液氮转运,冷冻保存在−80℃冰箱中。
1.1.2 入组和排除标准
入组标准:(1)行根治性食管癌切除术;(2)术前未接受过抗肿瘤治疗,包括放化疗、免疫及靶向等治疗;(3)术后病理检查明确诊断为ESCC;(4)临床病理资料完整。排除标准:(1)既往确诊严重的心脑血管疾病、肝肾功能衰竭、呼吸衰竭等重大疾病史;(2)合并其他肿瘤;(3)合并血液系统疾病;(4)围手术期死亡。
课题已通过我院伦理委员会批准并备案,患者及家属均签署自愿捐献知情同意书。
1.2 主要试剂及仪器
WISP-1多克隆抗体(18166-1-AP)和VEGF-A多克隆抗体(19003-1-AP)购自武汉三鹰公司,引物序列见表1。
表 1 引物序列Table 1 Primer sequencePrimers Primer sequence (5ʹ-3ʹ) WISP-1 Forword: GAGAGGTGGTCGGATCCTCT Reverse: CCAGTGGAGCTGGGGTAAAG VEGFA Forword: GCTCGGTGCTGGAATTTGAT Reverse: TCACTCACTTTGCCCCTGTC GAPDH Forword: GCACCGTCAAGGCTGAGAAC Reverse: TGGTGAAGACGCCAGTGGA 1.3 免疫组织化学实验
1.3.1 实验步骤
4 µm厚蜡块进行脱蜡、水化后使用高压蒸汽法修复抗原,滴加山羊血清,然后滴加适量的一抗稀释液[WISP-1(1∶100),VEGF-A(1∶100)],4℃湿盒中孵育过夜,滴加适量二抗,DAB显色,苏木精对比染色,切片脱水封片。
1.3.2 免疫组织化学判读标准
在双盲的情况下由专业的病理科医师阅片。随机挑选3个高倍镜视野,根据染色强度和阳性细胞所占百分比综合评判蛋白的表达情况。采用Mattern积分法作为判读WISP-1及VEGF-A蛋白的评分标准[4]:(1)细胞染色强度评分:无染色计0分,弱染色计1分,中度染色计2分,强染色计3分;(2)阳性细胞所占百分比评分:<25%计0分,25%~50%计1分,51%~75%计2分,≥75%计3分。WISP-1及VEGF-A蛋白表达情况的最终评分为细胞染色强度与阳性细胞所占百分比两种评分值的乘积,分值≥3分则评为阳性,<3分则评为阴性。
1.4 Real-time PCR实验
RNA的提取:RNA浓度在200~1 000 ng/µl,OD260/OD280值在1.8~2.1之间。cDNA合成:将样本RNA稀释到1 000 ng/µl,按Thermo公司cDNA合成试剂盒步骤操作。RT-PCR:配置20 µl反应体系。反应条件:预变性95℃5 min 1个循环;PCR反应95℃15 s,60℃30 s 40个循环;延伸60℃2 min 1个循环。RT-PCR结果分析:根据算法2−ΔΔct计算目的基因相对表达量。具体计算方式:ΔCt=Ct目的基因−Ct内参基因 ,ΔΔCt=ΔCt实验组−ΔCt对照组。
1.5 预后评价
从医院信息管理系统中查询入组患者的联系方式,以患者手术日期作为随访的开始日期,患者的死亡日期作为随访的终点。随访时间截止到2022年6月1日,比较不同组别患者总生存期(Overall survival, OS)和中位生存时间(Median overall survival, mOS)。OS为患者手术日期到患者死亡日期或随访截止日期,随访期间电话联系3次未接通定义为失访。
1.6 统计学方法
用SPSS 25.0软件对收集的数据进行统计学处理分析。计数资料采用卡方检验或Fisher确切概率法,符合正态分布的计量资料采用配对样本t检验,Pearson法对WISP-1与VEGF-A mRNA的表达水平进行相关性分析。对于患者OS的差异分析采用Kaplan-Meier法绘制生存曲线,Log rank检验对两组间差异进行比较,将单因素分析有意义的变量纳入Cox回归模型行多因素分析。P<0.05为差异有统计学意义。
2 结 果
2.1 癌组织与癌旁组织中WISP-1及VEGF-A蛋白的表达
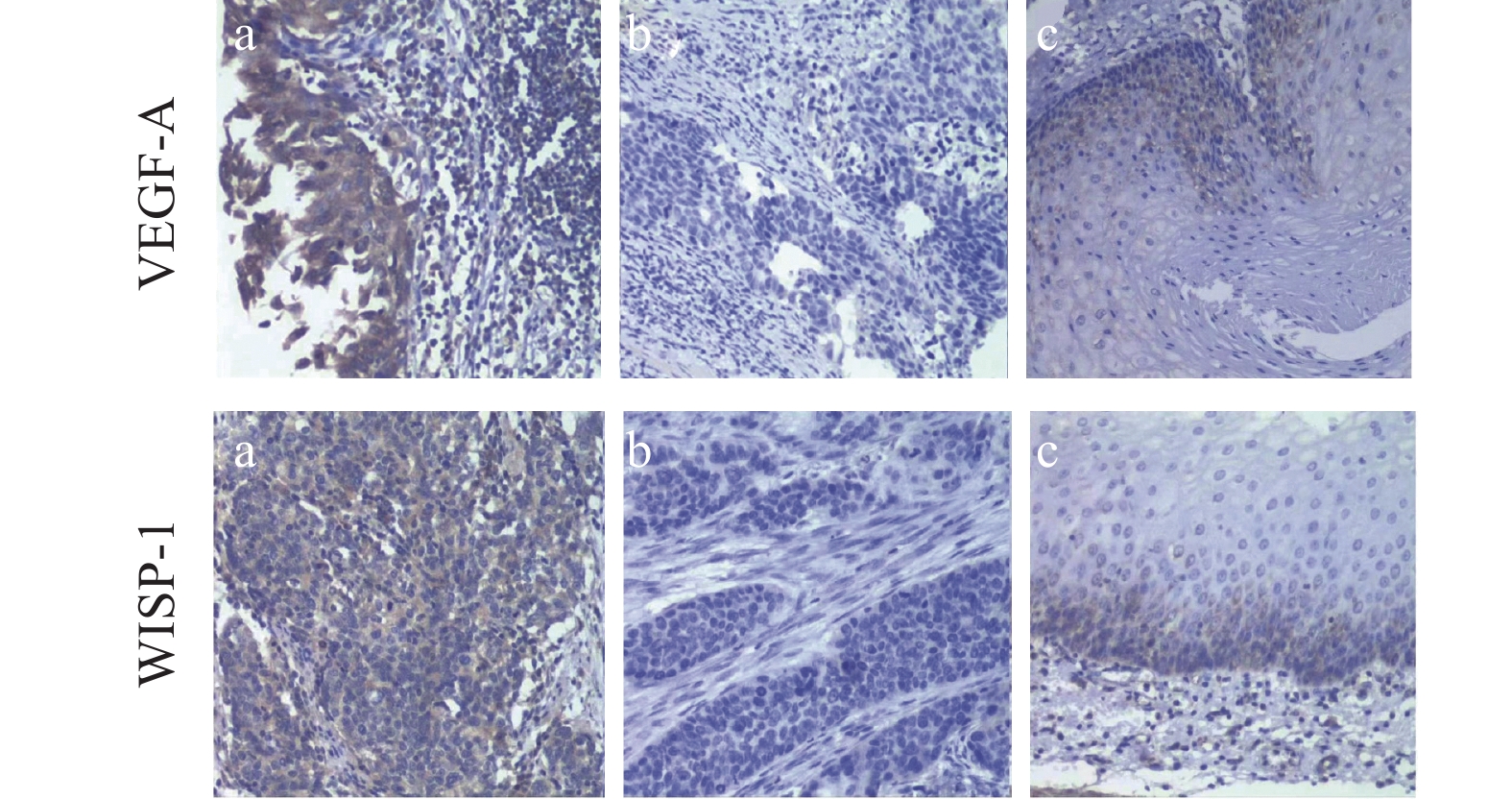
免疫组织化学染色结果显示WISP-1蛋白定位于细胞质,呈棕黄色,其在老年ESCC组中阳性表达率高于癌旁组,两者比较差异有统计学意义(P<0.05)。VEGF-A蛋白定位于细胞质,呈棕黄色,其在老年ESCC阳性表达率明显高于癌旁组织中的阳性表达率,差异具有统计学意义(P<0.001),见表2、图1。
表 2 WISP-1及VEGF-A在老年ESCC组织中的表达Table 2 Expression levels of WISP-1 and VEGF-A in elderly patients with ESCCGroup N WISP-1(n(%)) P VEGF-A(n(%)) P Positive Negative Positive Negative Cancerous tissue 90 48(53.33) 42(46.67) 0.04 45(50.00) 45(50.00) 0.001 Para-cancer tissues 48 17(35.42) 31(64.58) 10(20.83) 38(79.17) ![]() 图 1 免疫组织化学法检测WISP-1及VEGF-A在老年ESCC组织及癌旁组织中的表达(SP ×200)Figure 1 WISP-1 and VEGF-A protein expression levels in ESCC and para-cancer tissues of elderly patients detected by immunochemistry (SP ×200)a: positive expression in ESCC tissues; b:negative expression in ESCC tissues; c: positive expression in para-cancer tissues.
图 1 免疫组织化学法检测WISP-1及VEGF-A在老年ESCC组织及癌旁组织中的表达(SP ×200)Figure 1 WISP-1 and VEGF-A protein expression levels in ESCC and para-cancer tissues of elderly patients detected by immunochemistry (SP ×200)a: positive expression in ESCC tissues; b:negative expression in ESCC tissues; c: positive expression in para-cancer tissues.2.2 癌组织中WISP-1及VEGF-A蛋白表达与临床病理特征的关系
ESCC患者WISP-1蛋白阳性表达率在70岁以上及有淋巴结转移者显著高于65~70岁及无淋巴结转移者,差异有统计学意义(均P<0.05);WISP-1蛋白阳性表达与性别、肿瘤大小、浸润深度、TNM分期、神经侵犯、脉管侵犯、分化程度均未发现相关性,差异无统计学意义(均P>0.05)。ESCC患者VEGF-A蛋白阳性表达率在T3~T4、有淋巴结转移及Ⅲ~Ⅳ期患者显著高于T1~T2、无淋巴结转移及Ⅰ~Ⅱ期患者,差异均具有统计学意义(均P<0.05);VEGF-A蛋白阳性表达与性别、年龄、肿瘤大小、神经侵犯、脉管侵犯、分化程度均未显示相关性,差异无统计学意义(P>0.05),见表3。
表 3 老年ESCC组织中WISP-1及VEGF-A蛋白的表达与临床病理特征的相关性Table 3 Correlation of WISP-1 and VEGF-A proteins expression levels in cancer tissues with clinicopathological features of elderly ESCC patientsClinicopathological features N WISP-1(n(%)) P VEGF-A(n(%)) P Positive Negative Positive Negative Gender 0.126 0.634 Male 66 32(48.5) 34(51.5) 32(48.5) 34(51.5) Female 24 16(66.7) 80(33.3) 13(54.2) 11(45.8) Age(years) 0.047 0.267 65-70 59 27(45.8) 32(54.2) 32(54.2) 27(45.8) >70 31 21(67.7) 10(32.3) 13(41.9) 18(58.1) Tumor size(cm) 0.621 0.192 ≤4 56 31(55.4) 25(44.6) 25(44.6) 31(55.4) >4 34 17(50.0) 17(50.0) 20(58.8) 14(41.2) Depth of infiltration 0.706 0.030 T1-T2 34 19(55.9) 15(44.1) 12(35.3) 22(64.7) T3-T4 56 29(51.8) 27(48.2) 33(58.9) 23(41.1) Lymph node metastasis 0.032 0.006 Positive 43 28(65.1) 15(34.8) 28(65.1) 15(34.9) Negative 47 20(42.6) 27(57.4) 17(36.2) 30(63.8) TNM stage 0.458 0.010 Ⅰ-Ⅱ 52 26(50.0) 26(50.0) 20(38.5) 32(61.5) Ⅲ-Ⅳ 38 22(57.9) 16(42.1) 25(65.8) 13(34.2) Vascular invasion 0.073 0.655 Positive 30 12(40.0) 18(60.0) 16(53.3) 14(46.7) Negative 60 36(60.0) 24(40.0) 29(48.3) 31(51.7) Neural invasion 0.114 0.827 Positive 33 14(42.4) 19(57.6) 17(51.5) 16(48.5) Negative 57 34(59.6) 23(40.4) 28(49.1) 29(50.9) Degree of differentiation 0.852 0.813 Poor 39 22(56.4) 17(43.6) 18(46.2) 21(53.8) Moderate 38 19(50.0) 19(50.0) 20(52.6) 18(47.3) Well 13 7(53.8) 6(46.2) 7(53.8) 6(46.2) 2.3 癌组织与癌旁组织中WISP-1 mRNA及VEGF-A mRNA的表达
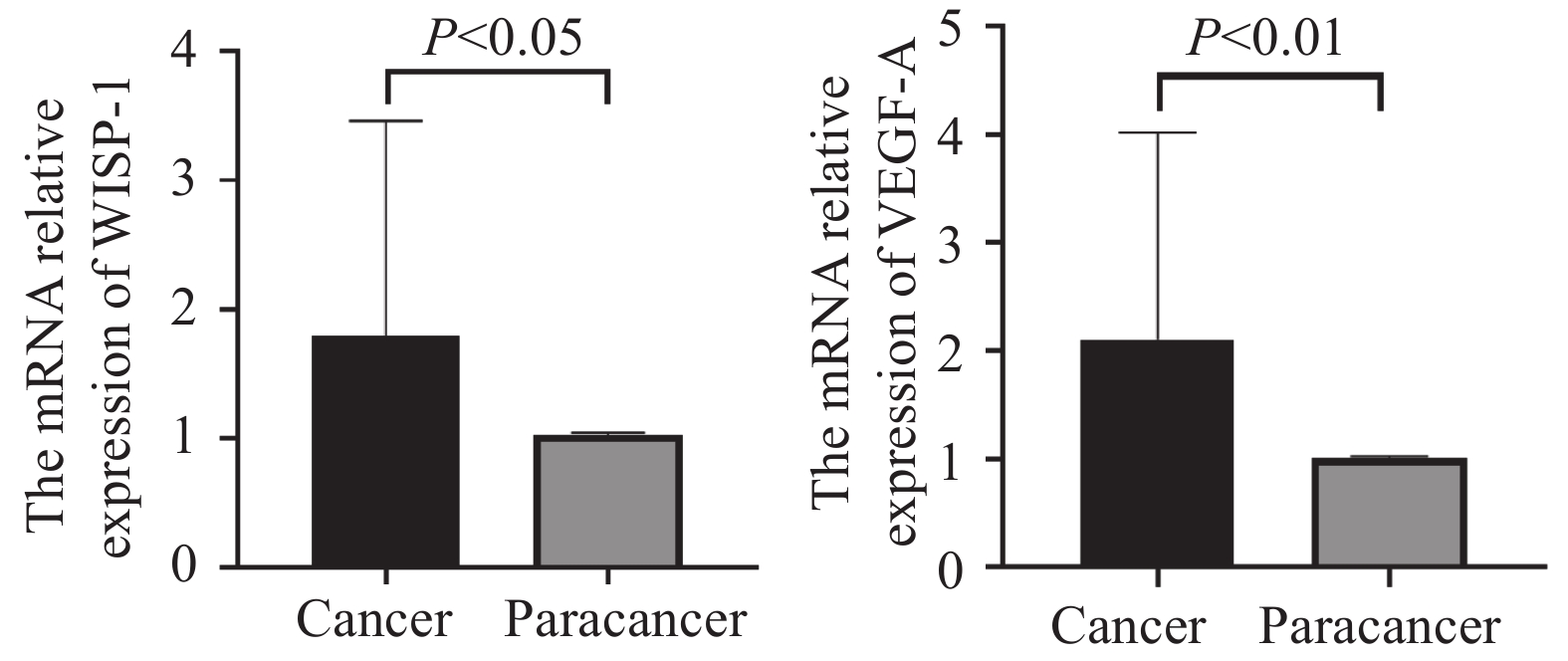
经正态性检验,30例患者样本mRNA表达的数据资料符合正态分布,因此采用配对样本t检验分析。WISP-1 mRNA在老年ESCC癌组织中表达量(1.793±1.663)显著高于癌旁配对组织(1.023±0.023),两组间差异有统计学意义(t=2.534,P<0.05);VEGF-A mRNA在老年ESCC癌组织(2.105±1.915)也显著高于癌旁配对组织(1.013±0.014),两组间差异有统计学意义(t=3.119,P<0.01),见图2。
2.4 WISP-1与VEGF-A在蛋白及转录水平的相关性分析
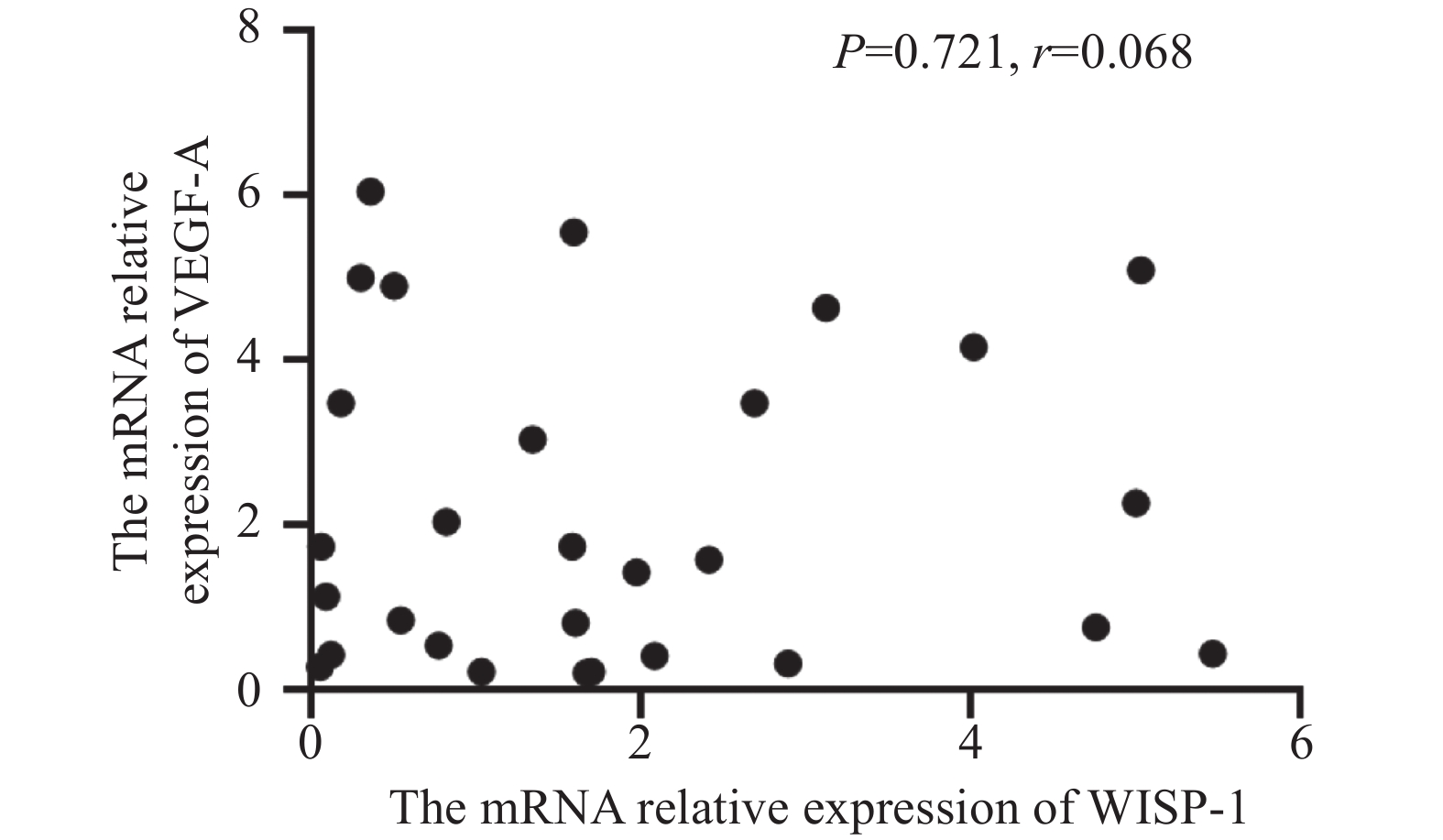
进一步分析WISP-1与VEGF-A的相关性。在90例老年ESCC病例中运用卡方检验分析两者在蛋白水平的相关性,结果显示WISP-1与VEGF-A的表达差异不具有统计学意义(χ2=0.179,P>0.05),见表4。在30例老年ESCC病例中运用Pearson相关性分析两者在转录水平的相关性,结果也未能显示二者在转录水平上有相关性(r=0.068,P>0.05),见图3,即与免疫组织化学结果分析一致。
表 4 WISP-1与VEGF-A蛋白表达的相关性Table 4 Correlation between WISP-1 and VEGF-A protein expression levelsWISP-1(+) WISP-1(-) χ2 P VEGF-A(+) 25(27.8%) 20(22.2%) 0.179 0.673 VEGF-A(-) 23(25.6%) 22(24.4%) 2.5 预后分析
本研究对90例患者进行随访,截至2022年6月1日,共获得85位患者的随访信息,其中存活患者38例,死亡患者47例,失访5例。OS为4~80个月,mOS为46个月。单因素分析结果表明WIPS1、VEGF-A阳性表达、肿瘤浸润深、有淋巴结转移、病理分期晚、肿瘤分化程度差的患者OS较短,预后更差(均P<0.05)。将上述单因素分析有统计学差异的指标引入多因素Cox风险回归模型中分析,结果显示只有淋巴结转移是影响老年ESCC患者预后的独立危险因素,差异具有统计学意义(P<0.001),见表5。
表 5 老年ESCC患者总体生存期的影响因素单因素及多因素分析Table 5 Univariate and multivariate Cox analyses of overall survival in elderly patients with ESCCPrognostic
indexN mOS
(months)Log rank
univariateMultivariate
Cox analysisχ2 P 95%CI P WISP-1 4.712 0.030 Positive 47 44 Negative 38 VEGF-A 7.299 0.007 Positive 43 43 Negative 42 Gender 0.641 0.423 Male 62 59 Female 23 44 Age
(years)0.002 0.967 65-70 56 51 >70 29 59 Tumor
size (cm)0.102 0.749 ≤4 53 51 >4 32 56 Depth of
infiltration5.384 0.020 T1-T2 33 T3-T4 52 43 Lymph node
metastasis20.591 <0.001 2.067-7.469 <0.001 Positive 42 31 Negative 43 TNM stage 18.055 <0.001 Ⅰ-Ⅱ 48 Ⅲ-Ⅳ 37 31 Vascular
invasion0.297 0.586 Positive 29 56 Negative 56 55 Neural
invasion1.783 0.182 Positive 30 31 Negative 55 59 Degree of
differentiation6.802 0.033 Poor 37 38 Moderate 35 Well 13 61 3 讨论
食管癌的发病率在40岁之后快速上升,随着我国人口老龄化,65岁以上年龄组的发病率明显增高[9],食管癌分期困难是导致其治疗效果欠佳的原因之一[10],寻找有效评估老年ESCC患者的分子标志物,对提高食管癌治疗效果及延长患者生存期均具有重要意义。
WISP-1在多种恶性肿瘤中高表达且与肿瘤的恶性行为相关。WISP-1与细胞增殖、凋亡、侵袭和转移相关[11],且已在结直肠癌、胃癌、肝癌[12-14]等明确上述结论。Nagai等使用IHC研究发现56%的ESCC患者出现WISP-1阳性表达,且WISP-1的表达与浸润深度、肿瘤大小、肿瘤类型、淋巴结转移及预后不良显著相关[7]。近期有文献报道78例不区分年龄的ESCC患者发现WISP-1的高表达提示ESCC患者淋巴结转移[15]。本研究用IHC检测90例老年ESCC蜡块发现癌组织中WISP-1阳性表达率显著高于癌旁组织,与Nagai等[7]研究结果基本一致。为使结果更具科学性,本课题进一步应用RT-PCR方法检测30例老年ESCC新鲜组织中WISP-1 mRNA的表达,结果显示其mRNA表达水平在ESCC也显著高于癌旁组织。因此,WISP-1在老年ESCC中可能同样参与癌的形成。本研究还发现WISP-1的表达水平在年龄超过70岁患者组显著高于65~70岁患者组,在有淋巴结转移患者高于淋巴结无转移的患者,提示年龄越大,有淋巴结转移者WISP-1表达越高。
VEGF-A在包括ESCC在内的人类各种癌症中过表达。本研究结果表明VEGF-A在老年ESCC中阳性表达率显著高于癌旁组织,且在转录水平同样得到了证实。VEGF-A阳性表达的老年ESCC更易有深的浸润和淋巴结转移。这一结果与Shao等[16]在不区分年龄ESCC中的研究结果基本一致。
WISP-1通过对VEGF-A表达的调节对肿瘤血管的生成产生一定的作用。在人类骨肉瘤中发现WISP-1通过FAK/JNK/HIF-1α信号通路,介导人骨肉瘤细胞VEGF-A的表达[3],提示WISP-1有可能是一种新的血管生成调节因子。到目前为止,尚未见有文献报道WISP-1与VEGF-A在ESCC患者中的相关性研究。本研究未发现老年ESCC中WISP-1与VEGF-A的表达具有相关性,提示WISP-1可能存在其他促进肿瘤侵袭和转移的途径,也可能是本研究样本量尚不足够大。
WISP-1高表达患者往往提示预后较差。Wang等发现WISP-1高表达是肝癌转移和预后不良的标志物[14]。Gaudreau等认为在多种实体瘤中WISP1高表达提示疾病晚期[17]。本研究Cox多因素回归分析未能显示WISP-1是老年ESCC患者的独立预后不良因素,但单因素分析中显示WISP-1阳性表达的OS和mOS均低于WISP-1阴性表达组,即WISP-1阳性表达的老年ESCC患者的预后较差。
VEGF-A阳性患者有可能生存期短于阴性患者。本研究发现VEGF-A阳性的患者OS和mOS明显低于VEGF-A阴性的患者,但多因素Cox回归分析未能证明VEGF-A是独立的危险因素,与Shi等和Shao等不区分年龄的ESCC研究基本一致[18-19]。但国内也有学者研究表明VEGF可能是在不区分年龄的ESCC预后独立影响因素[20]。出现上述结果差异可能是因为ESCC的异质性明显,也可能是年龄的因素,且研究多为回顾性研究,因此进一步深入研究可能会得出更有意义的结论。
综上所述,本研究应用IHC及RT-PCR的方法检测老年ESCC组织WISP-1与VEGF-A的表达,均显示两个分子在癌组织中的表达水平明显高于癌旁组织,且年龄越大,WISP-1表达越高,WISP-1和VEGF-A阳性表达者的预后较差。提示WISP-1和VEGF-A与老年ESCC的恶性行为相关,具有作为老年ESCC分子标志物的潜能。
Competing interests: The authors declare that they have no competing interests.利益冲突声明:所有作者均声明不存在利益冲突。作者贡献:李晨露:实验实施、数据分析、文献检索及论文撰写黄俊星:研究设计、选题、论文指导及修改 -
表 1 引物序列
Table 1 Primer sequence
Primers Primer sequence (5ʹ-3ʹ) WISP-1 Forword: GAGAGGTGGTCGGATCCTCT Reverse: CCAGTGGAGCTGGGGTAAAG VEGFA Forword: GCTCGGTGCTGGAATTTGAT Reverse: TCACTCACTTTGCCCCTGTC GAPDH Forword: GCACCGTCAAGGCTGAGAAC Reverse: TGGTGAAGACGCCAGTGGA 表 2 WISP-1及VEGF-A在老年ESCC组织中的表达
Table 2 Expression levels of WISP-1 and VEGF-A in elderly patients with ESCC
Group N WISP-1(n(%)) P VEGF-A(n(%)) P Positive Negative Positive Negative Cancerous tissue 90 48(53.33) 42(46.67) 0.04 45(50.00) 45(50.00) 0.001 Para-cancer tissues 48 17(35.42) 31(64.58) 10(20.83) 38(79.17) 表 3 老年ESCC组织中WISP-1及VEGF-A蛋白的表达与临床病理特征的相关性
Table 3 Correlation of WISP-1 and VEGF-A proteins expression levels in cancer tissues with clinicopathological features of elderly ESCC patients
Clinicopathological features N WISP-1(n(%)) P VEGF-A(n(%)) P Positive Negative Positive Negative Gender 0.126 0.634 Male 66 32(48.5) 34(51.5) 32(48.5) 34(51.5) Female 24 16(66.7) 80(33.3) 13(54.2) 11(45.8) Age(years) 0.047 0.267 65-70 59 27(45.8) 32(54.2) 32(54.2) 27(45.8) >70 31 21(67.7) 10(32.3) 13(41.9) 18(58.1) Tumor size(cm) 0.621 0.192 ≤4 56 31(55.4) 25(44.6) 25(44.6) 31(55.4) >4 34 17(50.0) 17(50.0) 20(58.8) 14(41.2) Depth of infiltration 0.706 0.030 T1-T2 34 19(55.9) 15(44.1) 12(35.3) 22(64.7) T3-T4 56 29(51.8) 27(48.2) 33(58.9) 23(41.1) Lymph node metastasis 0.032 0.006 Positive 43 28(65.1) 15(34.8) 28(65.1) 15(34.9) Negative 47 20(42.6) 27(57.4) 17(36.2) 30(63.8) TNM stage 0.458 0.010 Ⅰ-Ⅱ 52 26(50.0) 26(50.0) 20(38.5) 32(61.5) Ⅲ-Ⅳ 38 22(57.9) 16(42.1) 25(65.8) 13(34.2) Vascular invasion 0.073 0.655 Positive 30 12(40.0) 18(60.0) 16(53.3) 14(46.7) Negative 60 36(60.0) 24(40.0) 29(48.3) 31(51.7) Neural invasion 0.114 0.827 Positive 33 14(42.4) 19(57.6) 17(51.5) 16(48.5) Negative 57 34(59.6) 23(40.4) 28(49.1) 29(50.9) Degree of differentiation 0.852 0.813 Poor 39 22(56.4) 17(43.6) 18(46.2) 21(53.8) Moderate 38 19(50.0) 19(50.0) 20(52.6) 18(47.3) Well 13 7(53.8) 6(46.2) 7(53.8) 6(46.2) 表 4 WISP-1与VEGF-A蛋白表达的相关性
Table 4 Correlation between WISP-1 and VEGF-A protein expression levels
WISP-1(+) WISP-1(-) χ2 P VEGF-A(+) 25(27.8%) 20(22.2%) 0.179 0.673 VEGF-A(-) 23(25.6%) 22(24.4%) 表 5 老年ESCC患者总体生存期的影响因素单因素及多因素分析
Table 5 Univariate and multivariate Cox analyses of overall survival in elderly patients with ESCC
Prognostic
indexN mOS
(months)Log rank
univariateMultivariate
Cox analysisχ2 P 95%CI P WISP-1 4.712 0.030 Positive 47 44 Negative 38 VEGF-A 7.299 0.007 Positive 43 43 Negative 42 Gender 0.641 0.423 Male 62 59 Female 23 44 Age
(years)0.002 0.967 65-70 56 51 >70 29 59 Tumor
size (cm)0.102 0.749 ≤4 53 51 >4 32 56 Depth of
infiltration5.384 0.020 T1-T2 33 T3-T4 52 43 Lymph node
metastasis20.591 <0.001 2.067-7.469 <0.001 Positive 42 31 Negative 43 TNM stage 18.055 <0.001 Ⅰ-Ⅱ 48 Ⅲ-Ⅳ 37 31 Vascular
invasion0.297 0.586 Positive 29 56 Negative 56 55 Neural
invasion1.783 0.182 Positive 30 31 Negative 55 59 Degree of
differentiation6.802 0.033 Poor 37 38 Moderate 35 Well 13 61 -
[1] 杨欢, 孙宛怡, 王建炳, 等. 中国食管癌病因学、筛查及早期诊断研究进展[J]. 肿瘤防治研究, 2022, 49(3): 169-175. [Yang H, Sun WY, Wang JB, et al. Research Progress of tiology, Screening and Early Diagnosis of Esophageal Cancer in China[J]. Zhong Liu Fang Zhi Yan Jiu, 2022, 49(3): 169-175.] doi: 10.3971/j.issn.1000-8578.2022.21.1033 Yang H, Sun WY, Wang JB, et al. Research Progress of tiology, Screening and Early Diagnosis of Esophageal Cancer in China[J]. Zhong Liu Fang Zhi Yan Jiu, 2022, 49(3): 169-175. doi: 10.3971/j.issn.1000-8578.2022.21.1033
[2] Cao W, Chen HD, Yu YW, et al. Changing profiles of cancer burden worldwide and in China: a secondary analysis of the global cancer statistics 2020[J]. Chin Med J (Engl), 2021, 134(7): 783-791. doi: 10.1097/CM9.0000000000001474
[3] Tsai HC, Tzeng HE, Huang CY, et al. WISP-1 positively regulates angiogenesis by controlling VEGF-A expression in human osteosarcoma[J]. Cell Death Dis, 2017, 8(4): e2750. doi: 10.1038/cddis.2016.421
[4] 董学易, 赵秀兰, 赵楠, 等. Wnt1诱导信号通路蛋白1在胃腺癌中的表达及其意义[J]. 天津医药, 2021, 49(8): 796-801, 897. [Dong XY, Zhao XL, Zhao N, et al. Expression and significance of Wnt1 induced signaling pathway protein 1 in gastric adenocarcinoma[J]. Tianjin Yi Yao, 2021, 49(8): 796-801, 897.] doi: 10.11958/20210404 Dong XY, Zhao XL, Zhao N, et al. Expression and significance of Wnt1 induced signaling pathway protein 1 in gastric adenocarcinoma[J]. Tianjin Yi Yao, 2021, 49(8): 796-801, 897. doi: 10.11958/20210404
[5] Yang CL, Li F, Zhou WB, et al. Knockdown of long non-coding RNA CCAT2 suppresses growth and metastasis of esophageal squamous cell carcinoma by inhibiting the β-catenin/WISP1 signaling pathway[J]. J Int Med Res, 2021, 49(5): 3000605211019938.
[6] Linde P, Mallmann M, Adams A, et al. Chemoradiation for elderly patients (≥65 years) with esophageal cancer: a retrospective single-center analysis[J]. Radiat Oncol, 2022, 17(1): 187. doi: 10.1186/s13014-022-02160-w
[7] Nagai Y, Watanabe M, Ishikawa S, et al. Clinical significance of Wnt-induced secreted protein-1(WISP-1/CCN4) in esophageal squamous cell carcinoma[J]. Anticancer Res, 2011, 31(3): 991-997.
[8] Rice TW, Gress DM, Patil DT, et al. Cancer of the esophagus and esophagogastric junction-Major changes in the American Joint Committee on Cancer eighth edition cancer staging manual[J]. CA Cancer J Clin, 2017, 67(4): 304-317. doi: 10.3322/caac.21399
[9] 孟娜, 俞浩, 罗鹏飞, 等. 1990—2019年江苏省居民食管癌疾病负担及其危险因素变化趋势分析[J]. 中国肿瘤, 2023, 32(3): 184-190. [Meng N, Yu H, Luo PF, et al. Trend of burden on esophageal cancer and epidemio-logical change of related risk factors in Jiangsu Province from 1990 to 2019[J]. Zhongguo Zhong Liu, 2023, 32(3): 184-190.] doi: 10.11735/j.issn.1004-0242.2023.03.A004 Meng N, Yu H, Luo PF, et al. Trend of burden on esophageal cancer and epidemio-logical change of related risk factors in Jiangsu Province from 1990 to 2019[J]. Zhongguo Zhong Liu, 2023, 32(3): 184-190. doi: 10.11735/j.issn.1004-0242.2023.03.A004
[10] 陈克能. 冷静看待食管癌保器官的热潮[J]. 肿瘤防治研究, 2024, 51(2): 75-79. [Chen KN. Take A Calm View on Heat Wave of Organ Preservation in Esophageal Cancer Treatment[J]. Zhong Liu Fang Zhi Yan Jiu, 2024, 51(2): 75-79.] doi: 10.3971/j.issn.1000-8578.2024.24.0046 Chen KN. Take A Calm View on Heat Wave of Organ Preservation in Esophageal Cancer Treatment[J]. Zhong Liu Fang Zhi Yan Jiu, 2024, 51(2): 75-79. doi: 10.3971/j.issn.1000-8578.2024.24.0046
[11] Liu Y, Song Y, Ye M, et al. The emerging role of WISP proteins in tumorigenesis and cancer therapy[J]. J Transl Med, 2019, 17(1): 28. doi: 10.1186/s12967-019-1769-7
[12] Zhu Y, Li W, Yang Y, et al. WISP1 indicates poor prognosis and regulates cell proliferation and apoptosis in gastric cancer via targeting AKT/mTOR signaling pathway[J]. Am J Transl Res, 2020, 12(11): 7297-7311.
[13] Li Y, Zhu Z, Hou X, et al. LncRNA AFAP1-AS1 Promotes the Progression of Colorectal Cancer through miR-195-5p and WISP1[J]. J Oncol, 2021, 2021: 6242798.
[14] Wang QY, Feng YJ, Ji R. High expression of WISP1 promotes metastasis and predicts poor prognosis in hepatocellular carcinoma[J]. Eur Rev Med Pharmacol Sci, 2020, 24(20): 10445-10451.
[15] 金丽艳, 姚娟. WISP1在食管鳞癌组织和细胞中的表达及对细胞侵袭迁移的影响[J]. 临床肿瘤学杂志, 2021, 26(8): 680-686. [Jin LY, Yao J. Expression of WISP1 in tissues and cells ofesophageal squamous cell carcinoma and its influence on cell invasion and migration[J]. Lin Chuang Zhong Liu Xue Za Zhi, 2021, 26(8): 680-686.] doi: 10.3969/j.issn.1009-0460.2021.08.002 Jin LY, Yao J. Expression of WISP1 in tissues and cells ofesophageal squamous cell carcinoma and its influence on cell invasion and migration[J]. Lin Chuang Zhong Liu Xue Za Zhi, 2021, 26(8): 680-686. doi: 10.3969/j.issn.1009-0460.2021.08.002
[16] Shao JB, Li Z, Zhang N, et al. Hypoxia-inducible factor 1α in combination with vascular endothelial growth factor could predict the prognosis of postoperative patients with oesophageal squamous cell cancer[J]. Pol J Pathol, 2019, 70(2): 84-90. doi: 10.5114/pjp.2019.87100
[17] Gaudreau PO, Clairefond S, Class CA, et al. WISP1 is associated to advanced disease, EMT and an inflamed tumor microenvironment in multiple solid tumors[J]. Oncoimmunology, 2019, 8(5): e1581545. doi: 10.1080/2162402X.2019.1581545
[18] Shi ZZ, Wang WJ, Chen YX, et al. The miR-1224-5p/TNS4/EGFR axis inhibits tumour progression in oesophageal squamous cell carcinoma[J]. Cell Death Dis, 2020, 11(7): 597. doi: 10.1038/s41419-020-02801-6
[19] Shao N, Han Y, Song L, et al. Clinical significance of hypoxia-inducible factor 1α, and its correlation with p53 and vascular endothelial growth factor expression in resectable esophageal squamous cell carcinoma[J]. J Cancer Res Ther, 2020, 16(2): 269-275. doi: 10.4103/jcrt.JCRT_781_19
[20] 安秀英, 胡锋超, 张晶, 等. 食管鳞癌组织CD137L、VEGF、E-cad表达水平与患者预后的关系[J]. 诊断病理学杂志, 2022, 29(9): 810-813, 818. [An XY, Hu FC, Zhang J, et al. Relationship between the expression levels of CD137L, VEGF and E-cad in esophageal squamous cell carcinoma and the prognosis of patients[J]. Zhen Duan Bing Li Xue Za Zhi, 2022, 29(9): 810-813, 818.] doi: 10.3969/j.issn.1007-8096.2022.09.007 An XY, Hu FC, Zhang J, et al. Relationship between the expression levels of CD137L, VEGF and E-cad in esophageal squamous cell carcinoma and the prognosis of patients[J]. Zhen Duan Bing Li Xue Za Zhi, 2022, 29(9): 810-813, 818. doi: 10.3969/j.issn.1007-8096.2022.09.007



 下载:
下载: