Influence of Local Tumor Factors and Radiotherapy Dose on Prognosis of Clinical Stage T1-4N0M0 Esophageal Squamous Cell Carcinoma
-
摘要:目的
探讨不同放疗剂量对接受根治性放(化)疗的cT1-4N0M0期食管鳞癌患者不同肿瘤局部因素亚组人群预后的影响。
方法对256例临床无转移食管鳞癌患者进行回顾性分析,明确肿瘤局部因素指标预测患者预后的最佳截断值,分析近期疗效与肿瘤局部因素相关指标的关系,多因素分析影响患者OS的独立性指标,最后进行亚组分析,确定从不同放疗剂量中获益的亚组人群。
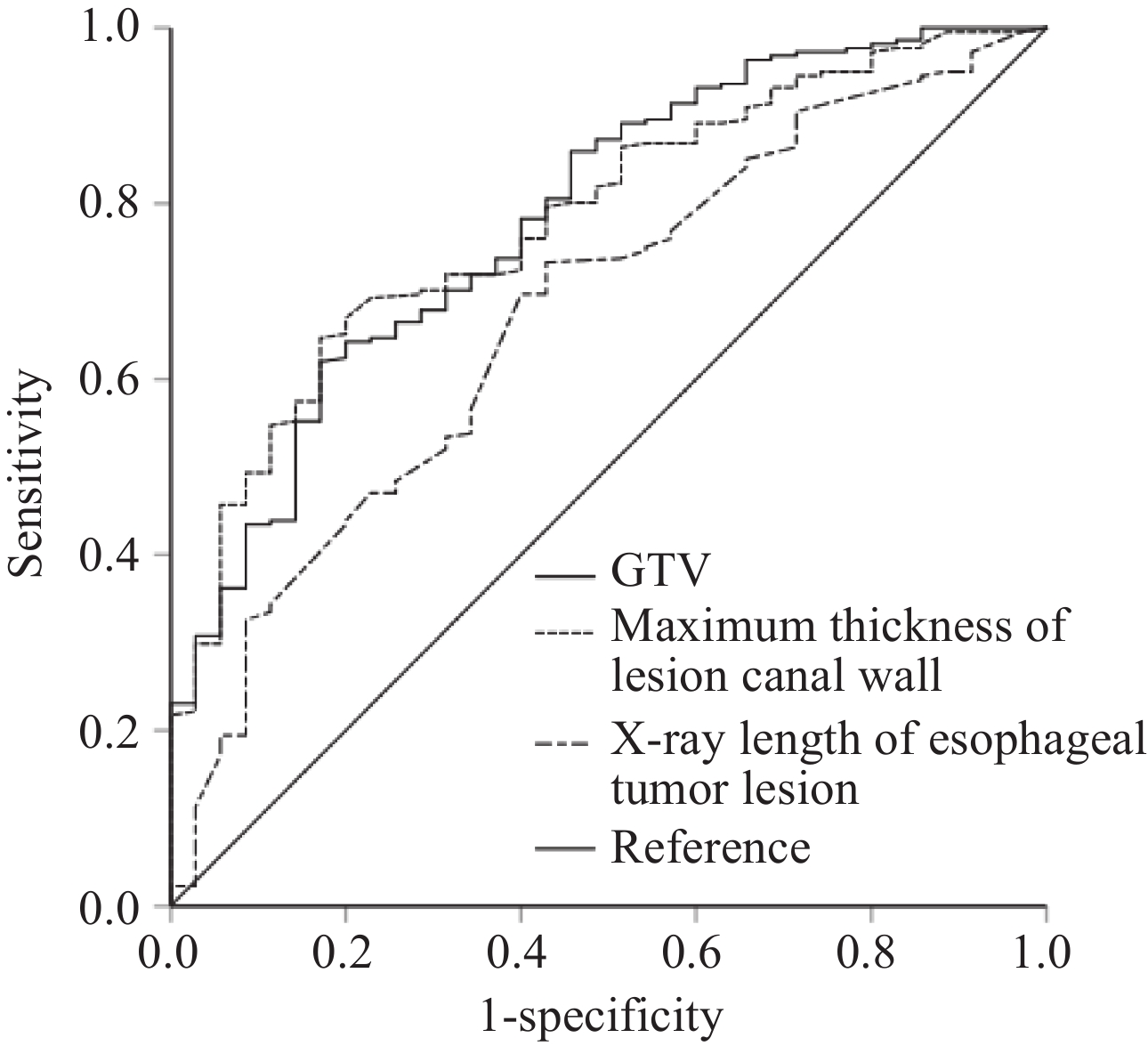
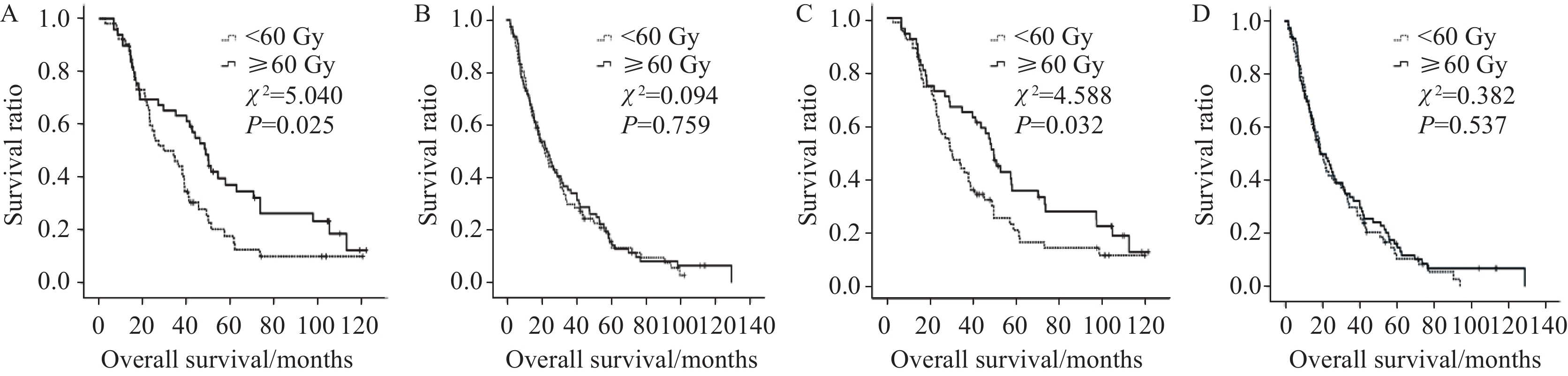
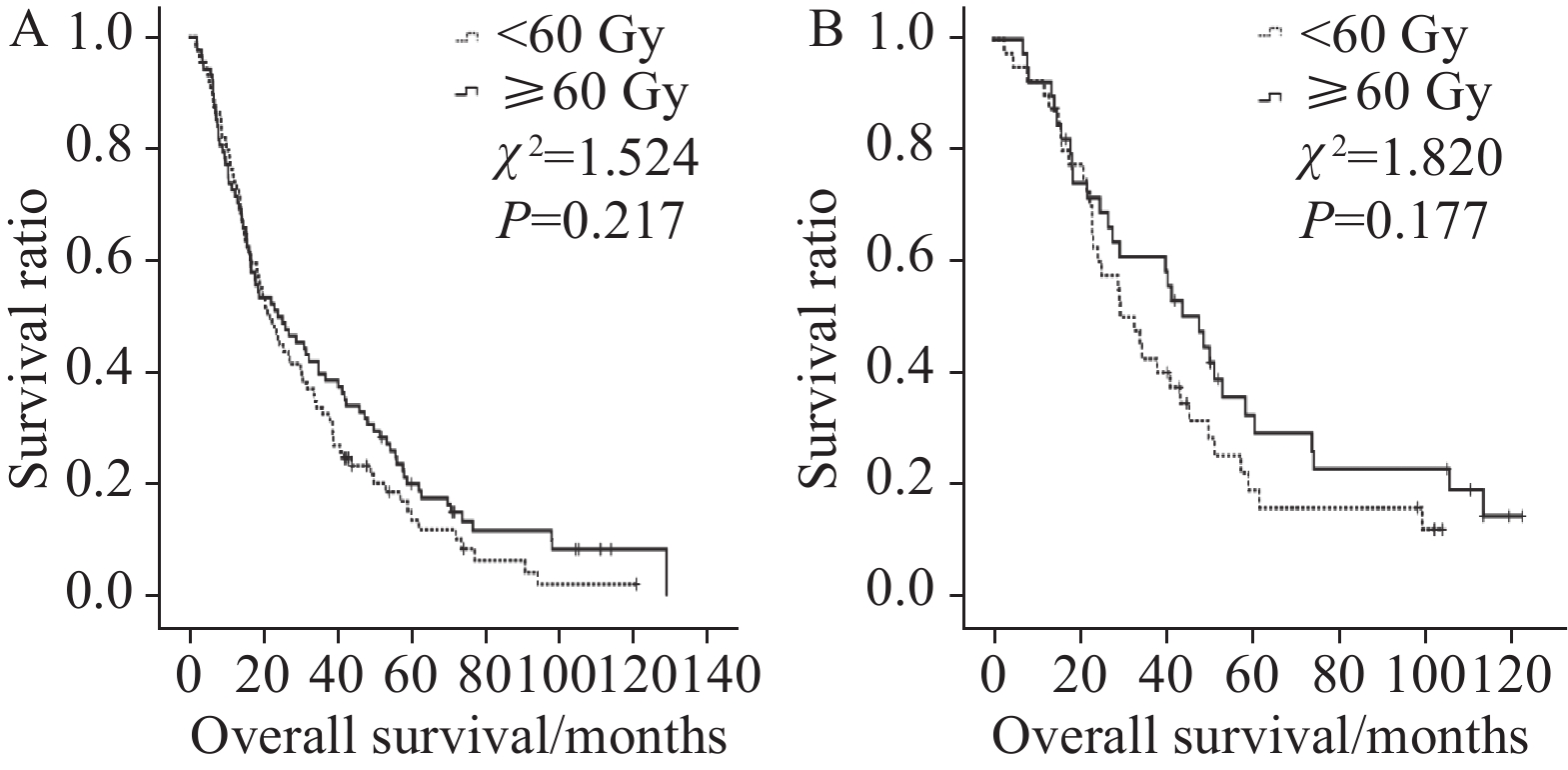
结果食管肿瘤病变X线长度越短、食管管壁厚度和GTV越小,患者近期疗效越好(χ2=9.066、10.310、15.661,P=0.011、0.006、<0.001)。多因素分析结果显示,食管GTV(P<0.001)、放疗剂量(P=0.038)和近期疗效(P<0.001)等3项指标为影响患者OS的独立性指标,后两项指标亦为影响患者PFS预后的独立性指标(P=0.033、P<0.001)。亚组分析结果显示放疗剂量较大(≥60 Gy)时,可使管壁最大厚度≤3.7 cm组、食管病变GTV≤37.34 cm3组和未接收同步化疗组患者OS(χ2=5.040、4.588、5.400,P=0.025、0.032、0.020)和PFS(χ2=6.089、4.353、6.459,P=0.014、0.037、0.011)均获益。
结论肿瘤局部因素指标为影响接受根治性放(化)疗食管鳞癌患者的重要预后因素;病变食管壁厚度和肿瘤体积较小的患者可能更能从较高的剂量(≥60 Gy)照射中获益。
Abstract:ObjectiveTo investigate the effect of different radiotherapy doses on the prognosis of patients with stage cT1-4N0M0 esophageal squamous cell carcinoma (ESCC) who received radical radio (chemo)therapy categorized into subgroups with different tumor local factors.
MethodsA retrospective analysis was conducted on 256 patients with clinically nonmetastatic esophageal squamous cell carcinoma. The optimal cutoff for tumor local factors was determined. The relationship between latest treatment efficacy and tumor local factors was analyzed, and independent indicators affecting patient overall survival (OS) were examined using multivariate analysis. The subgroup analysis was performed to determine the correlation between selected factors and radiation therapy doses.
ResultsThe shorter the X-ray length of esophageal tumor lesion and the smaller the lesion canal wall thickness and gross tumor volume (GTV), the better the latest treatment efficacy of the patients (χ2=9.066, 10.310, 15.661, respectively, P=0.011, 0.006, P<0.001). Multivariate analysis results showed that GTV(P<0.001), radiation dose(P=0.038), and latest treatment efficacy(P<0.001) were independent predictors of the patients’ OS, and the latter two were also independent predictors of the patients’ progression-free survival (PFS) (P=0.033, <0.001). Subgroup analysis further showed that high doses of radiotherapy (over 60 Gy) resulted in good OS (χ2=5.040, 4.588, 5.400, P=0.025, 0.032, 0.020) and PFS (χ2=6.089, 4.353, 6.459, P=0.014, 0.037, 0.011) in the subgroup with maximum wall thickness below 3.7 cm, with esophageal lesions with GTV below 37.34 cm3, or not receiving simultaneous chemotherapy.
ConclusionLocal tumor factors are important prognostic factors of ESCC patients treated with radical radio(chemo)therapy. Patients with thin lesion walls and small tumor volumes may greatly benefit from high doses of radiation (over 60 Gy).
-
0 引言
乳腺癌是全球范围内女性发病率最高的一种恶性肿瘤,在女性癌症相关死因中居第二位[1]。中国乳腺癌的发病率占世界总发病率的12.2%,死亡率高达9.6%,严重危害我国女性的身体健康和生活质量[2]。三阴性乳腺癌(triple negative breast cancer, TNBC)是指雌激素受体(ER)、孕激素受体(PR)和人类表皮生长因子受体2(HER2)均呈阴性表达的乳腺癌,恶性程度高,容易发生转移[3-4]。虽然目前对乳腺癌的诊断及治疗已取得一定成效,但三阴性乳腺癌的患者在1~3年内极易复发,并且大多于治疗后的5年内死亡[5]。针对以上问题,快速、准确地找到TNBC更有效的生物靶标成为关键所在。
环状RNA(circRNA)是生物体内广泛存在的一类具有调节功能的非编码RNA[6]。它们一般头尾连接形成环状结构,因为不含5' 末端帽和3' 末端poly(A)尾而表达稳定[7-8]。第一个环状RNA ciRS-7(CDR1as)由Hansen等首次发现,其可作为miR-7海绵在大脑中稳定表达并发挥重要的基因调控功能[9]。最近有研究发现,ciRS-7敲除后的体内动物模型出现了miR-7和miR-671表达失调,最终影响哺乳动物的大脑发育功能[10]。ciRS-7主要通过miRNA海绵的方式调节miR-7的表达,进而影响肿瘤的发生发展过程[9]。越来越多的研究发现环状RNA可调节人类多种肿瘤的进展。三阴性乳腺癌作为一类高度恶性的肿瘤,其潜在的分子机制仍有待阐明,而且环状RNA对其发病的调控作用尚未见报道。
本研究旨在探讨环状RNA ciRS-7在三阴性乳腺癌(TNBC)中的表达及其对细胞侵袭和迁移的影响,为寻找TNBC新型肿瘤生物标志物和治疗靶标奠定基础。
1 资料与方法
1.1 临床资料
选取2016年9月—2017年10月在河北医科大学第四医院接受手术治疗的乳腺癌患者132例,其中Luminal型89例、TNBC25例和HER2过表达型18例。所有患者均未接受术前化疗和放疗。所有肿瘤组织均由经验丰富的病理学专家证实。所有患者均签署了知情同意书,研究方案征得医院伦理委员会批准。
1.2 细胞株及主要试剂
人乳腺癌细胞株MDA-MB-231、MDA-MB-453、BT-549和MCF-7购自美国ATCC细胞库。RPMI 1640培养基、胰蛋白酶购自美国Gibco公司,胎牛血清购自上海依科赛生物制品有限公司。TRIzol购自美国Invitrogen公司,Platinum SYBR Super Mix试剂、Fu GENE HD转染试剂及反转录试剂盒均购自美国Promega公司,PCR引物购自英潍捷基贸易有限公司。Transwell小室(孔径3.0 μm)购自美国Corning公司,Matrigel基质胶购自美国BD公司。ciRS-7 siRNA购自广州锐博生物科技公司。
1.3 qRT-PCR法检测ciRS-7在乳腺癌组织和细胞中的表达
乳腺癌组织和细胞用TRIzol裂解后提取总RNA,按照Promega反转录试剂盒说明书制备cDNA,然后进行实时荧光定量PCR扩增,以GAPDH为内参,环状RNA ciRS-7来源于CDR1as,于是本实验设计两种引物,一种为发散引物,可扩增出环状转录本ciRS-7,另一种为聚合引物,能扩增CDR1as的线性转录本。然后以任意两例TNBC组织和两种TNBC细胞系(MDA-MB-231和BT-549)的cDNA和基因组DNA(gDNA)为模板,进行普通PCR反应。目的基因的引物序列及反应条件见表 1。利用各实验组目的基因及内参基因的Ct值,按照公式ΔCt=Ct目的基因-Ct内参基因,以2-ΔΔCt法计算目的基因的相对表达量。
表 1 PCR引物序列及反应条件Table 1 PCR primer sequences and reaction requirement
1.4 RT-PCR法检测ciRS-7在TNBC细胞中的表达
如1.3提取细胞总RNA后反转录为cDNA,PCR扩增,GAPDH为内参,目的基因的引物序列及反应条件,见表 1。扩增产物经2%琼脂糖凝胶电泳分离后进行核酸显色。
1.5 细胞转染
人TNBC细胞株MDA-MB-231和BT-549用含10%胎牛血清、100 u/ml青霉素和100 μg/ml链霉素的RPMI 1640培养液培养,于37℃、5%CO2体积分数的培养箱中。按Fu GENE HD转染试剂说明书方法将ciRS-7 siRNA转染至MDA-MB-231和BT-549细胞中,并在两种细胞中分别转染control siRNA作为对照。
1.6 划痕实验检测敲低ciRS-7后的细胞迁移能力
收集对数期的MDA-MB-231和BT-549细胞,调整细胞密度为5×105个每毫升,取2 ml接种于6孔板,并在6孔板背面划5条平行线做标记,24 h后用200 μl枪头在细胞中划2条垂直于背面平行线的直线,PBS冲洗2次,加入2 ml无胎牛血清的RPMI 1640培养液,于0、24 h在倒置显微镜下观察细胞向划痕中间迁移的距离并拍照,每组实验设3个平行孔。
1.7 Transwell小室检测敲低ciRS-7后的细胞迁移和侵袭能力
取对数期生长的细胞悬液,加入Transwell小室的上室(2×105个/小室),小室上铺胶检测侵袭能力,不铺胶者检测迁移能力; 下室加入600 μl含10%胎牛血清的RPMI 1640培养液,常规培养48 h,用棉签擦净上室细胞,PBS清洗后,结晶紫染色,倒置相差显微镜下(×200)观察并拍照,随机选取5个视野,计数穿膜细胞数。
1.8 动物实验
所有动物实验均经河北医科大学第四医院动物保护委员会批准。选用4周龄BALB/c裸鼠,随机分为2组(每组8只)。用si ciRS-7或对照siRNA转染的MDA-MB-231细胞(每只小鼠5×106个细胞)分别经尾静脉注射BALB/ c裸鼠中。然后将50 OD胆固醇修饰的siciRS-7或对照siRNA经尾静脉注射到BALB/c裸鼠中(每周2次)。2月后处死裸鼠,解剖肺组织。肉眼检查肺的外观,然后进行HE染色,并在显微镜下观察每个视野肺组织切片的转移克隆数。
上述实验均重复3次。
1.9 统计学方法
本实验数据应用SPSS21.0软件进行统计分析,计量资料以均数±标准(x±s)表示,两组之间均数比较采用t检验,P < 0.05为差异有统计学意义。
2 结果
2.1 环状RNA ciRS-7在TNBC组织和细胞中稳定高表达
结果显示,环状转录本ciRS-7只能用发散引物在cDNA中检测到,gDNA中未见扩增产物; 但是线性形式的CDR1as在cDNA和gDNA中均出现了扩增产物,见图 1A、B。这说明本研究使用的环状RNA ciRS-7引物的有效性,并进一步验证了其在TNBC中具有一定的表达丰度。qRT-PCR检测结果显示,TNBC样本中ciRS-7的相对表达量为(6.52±0.38),显著高于Luminal型(1.56±0.17)(P < 0.001)和HER2过表达型乳腺癌组织(2.27±0.66)(P < 0.001)以及癌旁正常组织(0.83±0.09)(P < 0.001),见图 1C。ciRS-7在TNBC细胞系MDA-MB-231和BT-549中高表达,但在Lunimal型MCF-7细胞和HER2过表达型MDA-MB-453细胞中低表达,见图 1D。qRT-PCR方法进一步证实ciRS-7对RNase R的抗性,MDA-MB-231和BT-549细胞经RNase R处理后CDR1线性基因表达明显下降,而环状形式的ciRS-7无明显变化,见图 1E。综上表明,环状RNA ciRS-7在TNBC组织和细胞中稳定高表达。
![]() 图 1 环状RNA ciRS-7在乳腺癌组织和细胞中的表达Figure 1 Expression of circular RNA ciRS-7 in breast cancer tissues and cell linesA: the expression of ciRS-7 in two cases of TNBC tissues detected by RT-PCR; B: the expression of ciRS-7 in MDA-MB-231 and BT-549 cells detected by RT-PCR; C: the expression of ciRS-7 in 132 cases of breast cancer tissue and paired normal adjacent tissues detected by qRT-PCR; D: the expression of ciRS-7 in MDA-MB-231, BT-549, MCF-7 and MDA-MB-453 cells detected by qRT-PCR; E: the expression of ciRS-7 with RNase R treatment in MDA-MB-231 and BT-549 cells detected by qRT-PCR; **: P < 0.01, ***: P < 0.001
图 1 环状RNA ciRS-7在乳腺癌组织和细胞中的表达Figure 1 Expression of circular RNA ciRS-7 in breast cancer tissues and cell linesA: the expression of ciRS-7 in two cases of TNBC tissues detected by RT-PCR; B: the expression of ciRS-7 in MDA-MB-231 and BT-549 cells detected by RT-PCR; C: the expression of ciRS-7 in 132 cases of breast cancer tissue and paired normal adjacent tissues detected by qRT-PCR; D: the expression of ciRS-7 in MDA-MB-231, BT-549, MCF-7 and MDA-MB-453 cells detected by qRT-PCR; E: the expression of ciRS-7 with RNase R treatment in MDA-MB-231 and BT-549 cells detected by qRT-PCR; **: P < 0.01, ***: P < 0.0012.2 环状RNA ciRS-7与乳腺癌患者临床病理特征的关系
为方便分析数据,将所有乳腺癌组织分类为ciRS-7高表达组和低表达组,使用132例乳腺癌患者组织中ciRS-7的平均相对表达量(4.82)作为所有乳腺癌患者的临界阈值。132例乳腺癌患者中ciRS-7高表达组32例,低表达组100例; ciRS-7表达与患者年龄、肿瘤直径和病理分级无关(均P > 0.05),而与分子分型、肿瘤浸润和淋巴结转移明显相关(均P < 0.05),见表 2。由此推断ciRS-7可能与乳腺癌患者的病情进展密切相关,于是本实验重点研究高表达ciRS-7且恶性度高的TNBC型乳腺癌。
表 2 ciRS-7表达与132例乳腺癌患者临床病理学指标的相关性Table 2 Correlation between ciRS-7 expression and clinicopathological parameters of 132 breast cancer patients
2.3 敲低ciRS-7可能抑制TNBC细胞的迁移能力
划痕实验结果显示siRNA介导的ciRS-7敲低组MDA-MB-231和BT-549细胞的迁移能力明显被抑制,见图 2A;转染ciRS-7 siRNA后两株细胞的ciRS-7表达明显降低(均P=0.000),见图 2B,证明本研究中针对环状RNA ciRS-7 siRNA干扰有效; 实验组和对照组两株细胞迁移面积变化百分率差异具有统计学意义,见图 2C(均P=0.000)。综上表明siRNA介导的ciRS-7敲低可能抑制TNBC细胞的迁移能力。
![]() 图 2 沉默ciRS-7对MDA-MB-231和BT-549细胞迁移的影响Figure 2 Effect of silencing ciRS-7 on migration of MDA-MB-231 and BT-549 cellsA: the migration of MDA-MB-231 and BT-549 cells in each group detected by wound healing assay; B: the expression of ciRS-7 after silencing ciRS-7 in MDA-MB-231 and BT-549 cells detected by qRT-PCR; C: the migration area in each group of MDA-MB-231 and BT-549 cells; ***: P < 0.001, compared with si ctrl group
图 2 沉默ciRS-7对MDA-MB-231和BT-549细胞迁移的影响Figure 2 Effect of silencing ciRS-7 on migration of MDA-MB-231 and BT-549 cellsA: the migration of MDA-MB-231 and BT-549 cells in each group detected by wound healing assay; B: the expression of ciRS-7 after silencing ciRS-7 in MDA-MB-231 and BT-549 cells detected by qRT-PCR; C: the migration area in each group of MDA-MB-231 and BT-549 cells; ***: P < 0.001, compared with si ctrl group2.4 敲低ciRS-7可能抑制TNBC细胞的迁移和侵袭能力
与si ctrl组相比,si ciRS-7处理组MDA-MB-231和BT-549细胞的穿膜数目明显减少,见图 3A。MDA-MB-231和BT-549中对照组和实验组的迁移和侵袭细胞数差异均有统计学意义(均P=0.000),见图 3B、C。
![]() 图 3 沉默ciRS-7对MDA-MB-231和BT-549细胞迁移和侵袭的影响Figure 3 Effect of silencing ciRS-7 on migration and invasion of MDA-MB-231 and BT-549 cellsA: the migration and invasion of MDA-MB-231 and BT-549 cells in each group detected by Transwell assay; B: the number of migrated and invaded MDA-MB-231 cells in each group; C: the number of migrated and invaded BT-549 cells in each group; ***: P < 0.001, compared with si ctrl group
图 3 沉默ciRS-7对MDA-MB-231和BT-549细胞迁移和侵袭的影响Figure 3 Effect of silencing ciRS-7 on migration and invasion of MDA-MB-231 and BT-549 cellsA: the migration and invasion of MDA-MB-231 and BT-549 cells in each group detected by Transwell assay; B: the number of migrated and invaded MDA-MB-231 cells in each group; C: the number of migrated and invaded BT-549 cells in each group; ***: P < 0.001, compared with si ctrl group2.5 敲低ciRS-7可能抑制裸鼠体内TNBC的肺转移
通过尾静脉注射建立MDA-MB-231细胞裸鼠肺转移模型。与si ctrl组相比,si ciRS-7组裸鼠的肺转移能力明显较弱,见图 4A。显微镜下观察si ciRS-7转染组的裸鼠形成的肺转移克隆数比si ctrl转染组的明显减少(P=0.000067),见图 4B。表明敲低ciRS-7可能抑制裸鼠体内TNBC的肺转移能力。
![]() 图 4 沉默ciRS-7对裸鼠体内TNBC的肺转移的影响Figure 4 Effect of silencing ciRS-7 on lung metastasis of TNBC in nude miceA: the metastasis clones of lung tissue in nude mice; Left: the appearance of lungs by naked eyes; Middle: HE staining(40×); Right: HE staining(100×); B: the number of clones for lung metastasis in each group; ***: P < 0.001, compared with si ctrl group
图 4 沉默ciRS-7对裸鼠体内TNBC的肺转移的影响Figure 4 Effect of silencing ciRS-7 on lung metastasis of TNBC in nude miceA: the metastasis clones of lung tissue in nude mice; Left: the appearance of lungs by naked eyes; Middle: HE staining(40×); Right: HE staining(100×); B: the number of clones for lung metastasis in each group; ***: P < 0.001, compared with si ctrl group3 讨论
本课题组检测了132例乳腺癌患者病理组织标本及配对癌旁正常组织中ciRS-7的表达水平,分析ciRS-7与乳腺癌患者临床病理特征之间的关系。另外检测了乳腺癌细胞系中ciRS-7的表达水平,通过体外细胞功能实验检测ciRS-7对TNBC细胞迁移和侵袭能力的影响,体内动物实验探究敲低ciRS-7后TNBC发生肺转移的变化,为寻找TNBC的新型肿瘤生物标志物和治疗靶标奠定基础。
由于当时研究技术的局限性,环状RNA在发现之初被认为是错误剪接或内含子套索过程中形成的副产物[11-13]。近年来随着RNA测序(RNA-seq)技术的不断发展,大量环状RNA在小鼠、线虫及人类细胞中被检测到[14-15]。circRNA的功能包括与AGO蛋白结合调控mRNA的转录翻译过程[6],与RNA结合蛋白相互作用参与转录后调控[16]。还有一类位于细胞核内的环状RNA通过抑制内含子环化正向调控其宿主基因的表达[17]。但目前研究最为深入的还是其作为miRNA分子“海绵”发挥生物学功能。
有报道称ciRS-7可作为miR-7的分子海绵参与多种癌症相关通路影响肿瘤的发生发展[18]。胃癌中ciRS-7表达上调,进而影响miR-7下游的PTEN/PI3K/AKT信号通路,促进癌症的发生发展[19]。食管鳞状细胞癌中过表达的ciRS-7可通过靶向miR-876-5p,解除其对肿瘤相关抗原MAGE-A家族的抑制作用从而加速ESCC进程[20]。结肠癌中ciRS-7可直接结合miR-7影响其下游的EGFR-RAF1信号通路,发挥关键的促癌作用[21]。ciRS-7还可通过充当miRNA分子海绵在非小细胞肺癌和膀胱癌的进展中发挥重要作用[22-23]。
然而,ciRS-7在乳腺癌中的研究未见报道。三阴性乳腺癌作为乳腺癌中具有高度转移潜能的分子类型,其调控机制至今尚未阐明。本研究采用qRT-PCR技术检测了4种乳腺癌细胞系中ciRS-7的表达水平,发现三阴性乳腺癌细胞系中ciRS-7的表达明显高于其他类型的乳腺癌细胞系,这提示ciRS-7可能与TNBC的高侵袭转移能力相关。
为此本课题组设计了细胞迁移和侵袭试验及裸鼠转移瘤实验进一步探究ciRS-7对TNBC的侵袭和转移能力的影响。结果表明环状RNA ciRS-7在体内和体外实验中均可促进TNBC细胞的侵袭迁移水平。但其具体的分子机制尚未研究透彻,猜测其很可能通过miRNA分子海绵机制发挥调控作用,这有待在今后的研究中得到证实。
Competing interests: The authors declare that they have no competing interests.利益冲突声明:所有作者均声明不存在利益冲突。作者贡献:孔雁、李曙光:实验设计、文章撰写董倩、许金蕊:数据分析、图片制作赵小涵、邓文钊:数据收集与整理沈文斌:研究指导、论文审阅、经费支持 -
图 3 不同放疗剂量对食管壁最大厚度≤3.7 cm(A)或>3.7 cm组(B)患者以及对食管病变GTV≤37.34 cm3(C)或>37.34 cm3组(D)患者预后影响生存曲线图
Figure 3 Kaplan-Meier plot of comparing patients treated with radiation dose below and over 60 Gy with maximum thickness of lesion canal wall ≤ 3.7 cm (A) or > 3.7 cm (B) and Kaplan-Meyer plot comparing patients treated with radiation dose below and over 60 Gy with GTV ≤37.34 cm3 (C) and > 37.34 cm3 (D)
表 1 肿瘤局部因素和患者预后ROC曲线分析结果
Table 1 Local tumor factors and ROC curve analysis for prognosis
Variable Minimum Maximum Median Mean Optimal cutoff AUC P Sensitivity Specificity X-ray length of esophageal
tumor lesion1.8 cm 15.7 cm 5.4 cm 5.7 cm 4.6 cm 0.674 0.001 73.3% 48.6% Maximum thickness of lesion
canal wall1.5 cm 8.5 cm 3.9 cm 4.0 cm 3.7 cm 0.782 <0.001 64.7% 82.9% GTV 6.74 cm3 265.21 cm3 39.48 cm3 51.86 cm3 37.34 cm3 0.784 <0.001 62.0% 83.6% 表 2 近期疗效与食管肿瘤局部因素之间关系的卡方检验分析结果 (n(%))
Table 2 Relation between recent treatment efficacy and local tumor factors (n (%))
Efficacy X-ray length of esophageal tumor lesion Maximum thickness of lesion canal wall GTV ≤4.6 cm >4.6 cm ≤3.7 cm >3.7 cm ≤37.34 cm3 >37.34 cm3 CR 34 (43.0) 45 (57.0) 41 (51.9) 38 (48.1) 46 (58.2) 33 (41.8) PR 43 (26.5) 119 (73.5) 58 (35.8) 104 (64.2) 66 (40.7) 96 (59.3) SD 2 (13.3) 13 (86.7) 2 (13.3) 13 (86.7) 1 (6.7) 14 (93.3) χ2 9.066 10.310 15.661 P 0.011 0.006 <0.001 表 3 影响患者预后的多因素分析结果
Table 3 Multivariate analysis of factors affecting patients’ prognosis
Factors Regression coefficient S Chi-square P OB 95%CI OS GTV 0.572 0.144 15.865 <0.001 1.771 1.337-2.346 Radiation dose −0.518 0.249 4.323 0.038 0.595 0.365-0.971 Recent treatment efficacy 0.611 0.135 20.425 <0.001 1.843 1.414-2.403 PFS Radiation dose −0.329 0.159 4.297 0.033 0.719 0.527-0.982 Recent treatment efficacy 0.748 0.158 22.541 <0.001 2.112 1.551-2.876 表 4 各项临床特征接受不同处方剂量的亚组分析结果
Table 4 Analysis of the effects of different radiation doses in subgroups
Variable Dose
(Gy)N OS (%) Median
(month)χ2 P PFS (%) Median
(month)χ2 P 3-year 5-year 3-year 5-year Gender Male <60 82 39.0 14.8 24.3 0.877 0.349 25.6 12.7 16.0 1.229 0.268 ≥60 82 47.0 21.0 31.1 39.0 17.1 20.0 Female <60 47 31.9 15.3 25.2 2.169 0.141 23.4 11.8 18.0 2.651 0.104 ≥60 45 44.4 28.4 29.4 37.8 20.0 23.1 Age (years) ≤69 <60 66 39.4 23.0 25.5 0.495 0.482 30.3 17.7 18.3 1.273 0.259 ≥60 69 52.2 28.0 41.4 44.9 22.6 30.0 >69 <60 63 33.3 6.8 23.0 2.956 0.086 19.0 7.1 18.0 2.289 0.103 ≥60 58 39.0 18.6 24.8 31.0 3.2 18.0 Location Cervical/upper thoracic <60 44 45.5 14.0 29.0 0.609 0.435 29.5 10.7 19.2 0.835 0.361 ≥60 35 48.6 17.1 32.4 40.0 17.1 27.7 Middle thoracic <60 64 29.7 18.4 23.0 1.983 0.159 21.9 14.7 14.6 2.110 0.146 ≥60 63 47.2 27.5 31.6 39.7 17.4 21.9 Lower thoracic <60 21 38.1 15.9 25.5 0.252 0.616 23.8 7.9 22.8 0.607 0.436 ≥60 29 41.4 24.1 26.0 34.5 20.7 23.1 X-ray length of esophageal tumor lesion (cm) ≤4.6 <60 40 42.5 18.8 29.5 1.820 0.177 27.5 11.3 18.0 3.507 0.061 ≥60 39 60.9 32.3 47.6 51.3 24.2 37.0 >4.6 <60 89 33.7 13.6 21.9 1.524 0.217 23.6 13.4 16.0 0.940 0.332 ≥60 88 39.8 20.1 24.0 33.0 15.6 18.3 Maximum thickness of lesion canal wall (cm) ≤3.7 <60 52 50.0 17.8 29.5 5.040 0.025 25.0 14.0 18.9 6.089 0.014 ≥60 49 63.3 37.2 48.7 55.1 27.1 39.0 >3.7 <60 77 29.9 13.3 21.9 0.094 0.759 24.7 11.3 15.2 0.210 0.647 ≥60 78 35.4 15.7 23.1 28.2 12.8 18.2 GTV (cm3) ≤37.34 <60 62 43.5 20.4 29.5 4.588 0.032 27.4 15.2 20.0 4.353 0.037 ≥60 51 64.7 36.1 49.8 52.9 23.9 37.4 >37.34 <60 67 29.9 10.5 20.0 0.382 0.537 22.4 10.1 14.6 0.843 0.359 ≥60 76 33.7 16.2 19.0 28.9 14.5 18.0 cT stage cT1+cT2 <60 40 42.5 26.0 29.5 0.666 0.414 27.5 21.7 20.0 0.460 0.498 ≥60 55 61.5 32.3 48.2 50.9 22.4 37.0 cT3 <60 28 42.9 15.3 27.2 0.333 0.564 32.1 10.7 20.0 0.088 0.767 ≥60 28 39.3 12.5 24.8 32.1 14.3 22.0 cT4 <60 61 29.5 7.4 19.2 1.162 0.281 19.7 6.6 12.9 2.737 0.098 ≥60 44 31.8 20.0 18.8 27.3 15.9 18.2 Chemotherapy No <60 97 29.9 11.3 23.0 5.400 0.020 22.7 9.3 15.8 6.459 0.011 ≥60 108 46.0 23.0 31.1 39.8 18.2 23.1 Yes <60 32 56.3 27.3 38.8 0.001 0.979 31.3 23.4 18.9 0.024 0.877 ≥60 19 47.4 27.6 26.0 31.6 18.9 14.0 -
[1] Park S, Oh D, Choi YL, et al. Durvalumab and tremelimumab with definitive chemoradiotherapy for locally advanced esophageal squamous cell carcinoma[J]. Cancer, 2022, 128(11): 2148-2158. doi: 10.1002/cncr.34176
[2] Wei T, Ti W, Song Q, et al. Study of PD-1 Inhibitors in Combination with Chemoradiotherapy/Chemotherapy in Patients with Esophageal Squamous Carcinoma[J]. Curr Oncol, 2022, 29(5): 2920-2927. doi: 10.3390/curroncol29050238
[3] Zhu H, Rivin Del Campo E, Ye J, et al. Involved-Field Irradiation in Definitive Chemoradiotherapy for Locoregional Esophageal Squamous Cell Carcinoma: Results From the ESO-Shanghai 1 Trial[J]. Int J Radiat Oncol Biol Phys, 2021, 110(5): 1396-1406. doi: 10.1016/j.ijrobp.2021.02.053
[4] Li M, Zhang X, Zhao F, et al. Involved-field radiotherapy for esophageal squamous cell carcinoma: theory and practice[J]. Radiat Oncol, 2016, 11: 18. doi: 10.1186/s13014-016-0589-7
[5] Chow R, Lock M, Lee SL, et al. Esophageal Cancer Radiotherapy Dose Escalation Meta Regression Commentary: "High vs. Low Radiation Dose of Concurrent Chemoradiotherapy for Esophageal Carcinoma With Modern Radiotherapy Techniques: A Meta-Analysis"[J]. Front Oncol, 2021, 11: 700300. doi: 10.3389/fonc.2021.700300
[6] Zhang W, Xie Q, Zhu B, et al. Intensity-modulated radiotherapy with more than 60 Gy improved the survival of inoperable patients with locally advanced esophageal squamous cell carcinoma: A population-based real-world study[J]. Medicine (Baltimore), 2022, 101(16): e29166. doi: 10.1097/MD.0000000000029166
[7] Huang BT, Huang RH, Zhang WZ, et al. Different definitions of esophagus influence esophageal toxicity prediction for esophageal cancer patients administered simultaneous integrated boost versus standard-dose radiation therapy[J]. Sci Rep, 2017, 7(1): 120. doi: 10.1038/s41598-017-00168-x
[8] Chen NB, Qiu B, Zhang J, et al. Intensity-Modulated Radiotherapy versus Three-Dimensional Conformal Radiotherapy in Definitive Chemoradiotherapy for Cervical Esophageal Squamous Cell Carcinoma: Comparison of Survival Outcomes and Toxicities[J]. Cancer Res Treat, 2020, 52(1): 31-40. doi: 10.4143/crt.2018.624
[9] Zhang J, Zhang W, Zhang B, et al. Clinical results of intensity-modulated radiotherapy for 250 patients with cervical and upper thoracic esophageal carcinoma[J]. Cancer Manag Res, 2019, 11: 8285-8294. doi: 10.2147/CMAR.S203575
[10] Hermann RM, Kober L, Christiansen H. Stereotactic body radiation therapy: radiobiological characteristics, physical-technical prerequisites, clinical applications[J]. Radiologe, 2021, 61(9): 853-862. doi: 10.1007/s00117-021-00903-6
[11] Shioyama Y, Onishi H, Takayama K, et al. Clinical Outcomes of Stereotactic Body Radiotherapy for Patients With Stage I Small-Cell Lung Cancer: Analysis of a Subset of the Japanese Radiological Society Multi-Institutional SBRT Study Group Database[J]. Technol Cancer Res Treat, 2018, 17: 1533033818783904.
[12] Kitagawa Y, Uno T, Oyama T, et al. Esophageal cancer practice guidelines 2017 edited by the Japan Esophageal Society: part 1[J]. Esophagus, 2019, 16(1): 1-24. doi: 10.1007/s10388-018-0641-9
[13] Shen WB, Zhu SC, Gao HM, et al. Analysis of failure patterns in patients with resectable esophageal squamous cell carcinoma receiving chemoradiotherapy[J]. J Cancer Res Ther, 2016, 12(1): 62-68. doi: 10.4103/0973-1482.146128
[14] Qin Q, Ge X, Wang X, et al. Stage Ⅲ Esophageal Squamous Cell Carcinoma Patients With Three-Dimensional Conformal or Intensity-Modulated Radiotherapy: A Multicenter Retrospective Study[J]. Front Oncol, 2020, 10: 580450. doi: 10.3389/fonc.2020.580450
[15] Jiang N, Ge XL, Zhang ZY, et al. Prognostic Factors for Patients with Esophageal Cancer Receiving Definitive Radiotherapy Alone: A Retrospective Analysis[J]. Cancer Manag Res, 2021, 13: 3229-3234. doi: 10.2147/CMAR.S300672
[16] Bütof R, Löck S, Soliman M, et al. Dose-volume predictors of early esophageal toxicity in non-small cell lung cancer patients treated with accelerated-hyperfractionated radiotherapy[J]. Radiother Oncol, 2020, 143: 44-50. doi: 10.1016/j.radonc.2019.11.002
[17] Zhao J, Lei T, Zhang T, et al. The efficacy and safety of simultaneous integrated dose reduction in clinical target volume with intensity-modulated radiotherapy for patients with locally advanced esophageal squamous cell carcinoma[J]. Ann Transl Med, 2020, 8(18): 1160. doi: 10.21037/atm-20-4366
[18] Tan L, Cheng D, Wen J, et al. Identification of prognostic hypoxia-related genes signature on the tumor microenvironment in esophageal cancer[J]. Math Biosci Eng, 2021, 18(6): 7743-7758. doi: 10.3934/mbe.2021384
[19] Zhou X, You M, Wang F, et al. Multifunctional Graphdiyne-Cerium Oxide Nanozymes Facilitate MicroRNA Delivery and Attenuate Tumor Hypoxia for Highly Efficient Radiotherapy of Esophageal Cancer[J]. Adv Mater, 2021, 33(24): e2100556. doi: 10.1002/adma.202100556
[20] Hulshof MCCM, Geijsen ED, Rozema T, et al. Randomized study on dose escalation in definitive chemoradiation for patients with locally advanced esophageal cancer (ARTDECO Study)[J]. J Clin Oncol, 2021, 39(25): 2816-2824. doi: 10.1200/JCO.20.03697
[21] Lin FC, Chang WL, Chiang NJ, et al. Radiation dose escalation can improve local disease control and survival among esophageal cancer patients with large primary tumor volume receiving definitive chemoradiotherapy[J]. PLoS One, 2020, 15(8): e0237114. doi: 10.1371/journal.pone.0237114
[22] Favareto SL, Sousa CF, Pinto PJ, et al. Clinical Prognostic Factors for Patients With Esophageal Cancer Treated With Definitive Chemoradiotherapy[J]. Cureus, 2021, 13(10): e18894.
[23] Matsumoto S, Wakatsuki K, Nakade H, et al. Impact of CT-assessed changes in tumor size after neoadjuvant chemotherapy on pathological response and survival of patients with esophageal squamous cell carcinoma[J]. Langenbecks Arch Surg, 2022, 407(3): 965-974. doi: 10.1007/s00423-022-02430-x
[24] Wongwaiyut K, Ruangsin S, Laohawiriyakamol S, et al. Pretreatment Esophageal Wall Thickness Associated with Response to Chemoradiotherapy in Locally Advanced Esophageal Cancer[J]. J Gastrointest Cancer, 2020, 51(3): 947-951. doi: 10.1007/s12029-019-00337-3
[25] Li TT, Fu XB, Xiao LH, et al. The long-term impact of tumor burden in pT3N0M0 esophageal squamous cell carcinoma: A propensity score-matched analysis[J]. Medicine (Baltimore), 2019, 98(42): e17637. doi: 10.1097/MD.0000000000017637
[26] Yue YT, Li N, Shahid H, et al. Gross Tumor Volume Definition and Comparative Assessment for Esophageal Squamous Cell Carcinoma From 3D 18 F-FDG PET/CT by Deep Learning-Based Method[J]. Front Oncol, 2022, 12: 799207. doi: 10.3389/fonc.2022.799207
[27] Chen Y, Huang Q, Chen J, et al. Primary gross tumor volume is prognostic and suggests treatment in upper esophageal cancer[J]. BMC Cancer, 2021, 21(1): 1130. doi: 10.1186/s12885-021-08838-w
[28] Huang RH, Guo H, Chen JZ, et al. Intratreatment Tumor Volume Change During Definitive Chemoradiotherapy is Predictive for Treatment Outcome of Patients with Esophageal Carcinoma[J]. Cancer Manag Res, 2020, 12: 7331-7339. doi: 10.2147/CMAR.S246500
[29] Li SH, Rau KM, Lu HI, et al. Pre-treatment maximal oesophageal wall thickness is independently associated with response to chemoradiotherapy in patients with T3-4 oesophageal squamous cell carcinoma[J]. Eur J Cardiothorac Surg, 2012, 42(6): 958-964. doi: 10.1093/ejcts/ezs136
[30] Wu Y, Li J. Change in Maximal Esophageal Wall Thickness Provides Prediction of Survival and Recurrence in Patients with Esophageal Squamous Cell Carcinoma After Neoadjuvant Chemoradiotherapy and Surgery[J]. Cancer Manag Res, 2021, 13: 2433-2445. doi: 10.2147/CMAR.S295646
[31] Chen CZ, Chen JZ, Luo T, et al. Late Toxicities, Failure Patterns, Local Tumor Control, and Survival of Esophageal Squamous Cell Carcinoma Patients After Chemoradiotherapy With a Simultaneous Integrated Boost: A 5-Year Phase Ⅱ Study[J]. Front Oncol, 2021, 11: 738936. doi: 10.3389/fonc.2021.738936
[32] Chen Y, Zhu HP, Wang T, et al. What is the optimal radiation dose for non-operable esophageal cancer? Dissecting the evidence in a meta-analysis[J]. Oncotarget, 2017, 8(51): 89095-89107. doi: 10.18632/oncotarget.18760
[33] Xu Y, Dong B, Zhu W, et al. A phase III multicenter randomized clinical trial of 60 Gy vs. 50 Gy radiation dose in concurrent chemoradiotherapy for inoperable esophageal squamous cell carcinoma[J]. Clin Cancer Res, 2022, 28(9): 1792-1799. doi: 10.1158/1078-0432.CCR-21-3843
[34] Fokas E, Martin D, Rödel C. Radiotherapy dose escalation does not improve local control in patients with esophageal cancer after definitive radiochemotherapy: the ARTDECO randomized phase Ⅲ trial[J]. Strahlenther Onkol, 2022, 198(4): 397-399. doi: 10.1007/s00066-022-01906-5
[35] Luo HS, Huang HC, Lin LX, et al. Effect of modern high-dose versus standard-dose radiation in definitive concurrent chemo-radiotherapy on outcome of esophageal squamous cell cancer: a meta-analysis[J]. Radiat Oncol, 2019, 14(1): 178. doi: 10.1186/s13014-019-1386-x



 下载:
下载: