ALDH5A1 Downregulation Promotes Tumor Metastasis and Contributes to Poor Prognosis in Ovarian Cancer
-
摘要:目的
探讨醛脱氢酶5A1(ALDH5A1)在卵巢癌组织中的表达、临床意义及其影响卵巢癌细胞侵袭转移的作用和分子机制。
方法首先基于GEO数据库比较ALDH5A1在卵巢癌转移组织和原发灶的表达差异。然后通过转染小干扰RNA(siRNA)方式建立ALDH5A1表达下调的卵巢癌细胞株SKOV3,划痕实验和Transwell侵袭实验检测细胞迁移侵袭能力的变化。cBioPortal数据库描绘了ALDH5A1在卵巢癌细胞株和卵巢癌患者中的共表达基因谱。最后运用TCGA和GEO数据库分析ALDH5A1表达水平与卵巢癌预后的相关性,HPA数据库再次确认卵巢癌患者中ALDH5A1和MMPs的相对表达水平。
结果相比于卵巢癌原发灶,转移组织中的ALDH5A1表达下调。细胞实验结果显示下调ALDH5A1表达可促进卵巢癌细胞株迁移和侵袭。富集分析发现ALDH5A1的共表达基因在细胞外基质(ECM)组织通路中显著富集,进一步通过数据库转录组数据验证ECM组织通路中发挥重要作用的基质金属蛋白酶(MMP)表达与ALDH5A1表达水平呈负相关,提示ALDH5A1可能通过ECM组织通路影响卵巢癌的转移和侵袭能力。生存分析结果提示ALDH5A1低表达的卵巢癌患者预后更差。
结论ALDH5A1表达下调可能促进卵巢癌侵袭转移,且与预后不良相关。
Abstract:ObjectiveTo investigate the expression level and clinical significance of ALDH5A1 in ovarian cancer (OC) tissues, as well as to explore the possible mechanism associated with the invasion and migration of OC cells.
MethodsWe initially compared ALDH5A1 expression in metastatic tissues and the primary site of OC based on the GEO database. Then, wound-healing and Transwell assays were utilized to determine the biological role of OC cells transfected with ALDH5A1 siRNA. To unravel the potential mechanism of ALDH5A1 meditating the metastasis of OC, the coexpression profile of ALDH5A1 in OC cell lines and OC patients were generated using cBioPortal. Moreover, the TCGA and GEO databases were used to analyze the relationship between ALDH5A1 expression and the prognosis of OC patients. The HPA database was further used to confirm the relative expression of ALDH5A1 and MMPs in OC patients.
ResultsALDH5A1 expression was downregulated in metastatic tissues compared with the primary site of OC, and ALDH5A1 knockdown promoted the malignant behavior of OC cells. Additionally, the coexpression profile of ALDH5A1 was significantly enriched in the extracellular matrix (ECM) organization pathway. Western blot assay further confirmed that the expression of MMP, which played an important role in the ECM pathway, was negatively correlated with ALDH5A1 expression in OC. These results indicated that ALDH5A1 may participate in the metastasis and invasion of OC via the ECM organization pathway. Finally, KM survival plots revealed that the survival rates of OC patients with lower ALDH5A1 expression were obviously lower.
ConclusionALDH5A1 downregulation may promote the tumor metastasis and contribute to poor prognosis in OC.
-
0 引言
卵巢癌是最常见的女性恶性肿瘤之一,据统计,2020年中国有约55 300例卵巢癌新发病例和37 500例死亡病例[1]。卵巢癌转移能力较强,多数于诊断时已出现疾病进展或广泛转移,患者5年生存率不足30%[2],因此,研究卵巢癌侵袭转移的机制对于该疾病的治疗具有重要意义。
近年来大量研究揭示了线粒体代谢相关基因与肿瘤转移、发展密切相关[3-5]。醛脱氢酶5家族A1(ALDH5A1)编码产物为琥珀酸半醛脱氢酶(SSADH),在线粒体代谢中通过催化琥珀酸半醛的氧化反应来降解γ-氨基丁酸(GABA)。ALDH5A1表达下调,SSADH功能缺陷会导致GABA正常降解途径受阻,转而生成γ-羟基丁酸(GHB),Hilvo等[6]报道了GHB可作为高级别卵巢浆液性癌生物标志,其蓄积提示卵巢癌的进展以及预后不良。
Deng等[7]发现ALDH5A1表达下调可通过抑制甲状腺癌细胞增殖、转移、侵袭、上皮间质转化等途径进而抑制肿瘤发生发展;Kaur等[8]采用Gene Sapiens微阵列数据库进行Meta分析发现,在特定类型肿瘤如胶质瘤、白血病、淋巴瘤中发现ALDH5A1表达上调,而在乳腺癌中ALDH5A1则较正常组织表达下调。
本课题组前期研究也表明,卵巢癌患者肿瘤组织ALDH5A1较正常卵巢组织表达下调,且ALDH5A1低表达与卵巢癌患者临床预后不良相关[9],然而具体分子机制尚不清楚。本研究将进一步探讨ALDH5A1在卵巢癌发生发展中的作用。
1 资料与方法
1.1 细胞
人卵巢癌上皮SKOV3细胞系来自美国ATCC细胞库。该细胞系在添加10%胎牛血清(FBS,购于美国Gibco公司)和1%青霉素/链霉素(购于美国Gibco公司)的McCoy’s 5A培养基(购于瑞士Lonza公司)中培养,置于37℃、5%CO2的加湿培养箱中。
1.2 生物信息数据库
GEO数据库[9]下载GSE20565卵巢癌转录组数据,分析ALDH5A1 mRNA表达水平。为进一步证实ALDH5A1和共表达基因的蛋白质组学表达,分析从人类蛋白图谱数据库(https://www.proteinatlas.org/)中获得的卵巢癌组织芯片(TMA)队列。
基于网络工具PROGgeneV2[10]评估卵巢癌中ALDH5A1表达水平与患者预后的相关性。运用TCGA数据库中ALDH5A1 mRNA表达数据和578例卵巢癌患者的总生存率信息建立Kaplan-Meier生存曲线图。Kaplan-Meier plotter在线平台芯片数据分析不同TP53突变型别与卵巢癌患者总生存率的关系。
1.3 实验方法
1.3.1 小干扰RNA
下调目的基因ALDH5A1 ALDH5A1 siRNA购自上海生工生物技术有限公司。靶标序列如下:siRNA1: 5’-CGGAAGTGGTACAATTTAATG-3’;siRNA2: 5’-GGTTCAACAACTACAGGAAAG-3’。LipofectamineTM3000(Thermo Fisher)将siRNA和阴性对照按说明书转染到SKOV3细胞中。转染48 h后测定敲减效率。
1.3.2 细胞划痕实验
SKOV3野生型细胞和转染siRNA的细胞按1×106个细胞/孔的密度接种于六孔板中,在5%CO2、37℃下孵育。过夜培养,细胞生长到100%密度。用移液器无菌100 μl枪头尖端在单层细胞上划出划痕。PBS冲洗细胞1次,去除细胞碎片,抚平划痕边缘,再用2.5 ml McCoy's 5A培养基进行细胞培养。倒置显微镜摄取划痕边缘的图像。
1.3.3 细胞侵袭实验
依据试剂盒说明书,用包被人工基质层(Matrigel)的Transwell小室评估SKOV3细胞侵袭能力。将基质在冰上融化,然后将30 μl的基质添加到24孔的Transwell小室中并凝固,野生或转染siRNA的细胞按1×105个细胞/孔的密度置于基质凝胶涂层顶部,在37℃、5%CO2下孵育10 min,使细胞下沉。下室为含有McCoy's 5A培养基,10%胎牛血清作为化学引诱剂。在5%CO2、37℃下孵育24 h后,用棉签去除未穿透膜的细胞。成功迁移至膜底面的细胞,4%聚甲醛固定,0.2%结晶紫染色10 min。倒置显微镜下计数细胞数量。
1.3.4 ALDH5A1共表达基因数据库检索与分析
来自Cancer Cell Line Encyclopedia(CCLE)中的卵巢癌细胞系(n=47)和TCGA中的卵巢浆液性囊腺癌(n=489)患者数据用cBioPortal[11]在线平台进行分析。当Spearman相关性 > 0.2和P < 0.05时,认为该基因为ALDH5A1共表达基因。采用g: Profiler[12]和Metascape[13]两个在线平台数据库中ALDH5A1的共同共表达基因进行通路富集分析。Metascape在线平台将具有相似功能的重要GO关键词可视化为交互网络,进一步确定关键词之间的关系,其中P < 0.01、相关度评分 > 0.3的项位于网状图边缘。此外,运用cBioPortal对ALDH5A1与细胞外基质(ECM)组织通路关键基因进行共表达分析。
1.3.5 实时荧光定量RT-PCR(qRT-PCR)
试剂盒(Invitrogen)提取细胞总RNA,用反转录酶(TransGen Biotech)以1 μg总RNA合成cDNA。SYBR Green PCR Master Mix(Takara Bio)和Light Cycler(Roche)行qRT-PCR分析。结果用2-∆∆ct计算。
1.4 统计学方法
所有数据使用GraphPad Prism 8.0进行分析,以三次重复的(x±s)表示。两组间定量资料用Student's t检验。P < 0.05为差异有统计学意义。
2 结果
2.1 ALDH5A1下调促进卵巢癌细胞侵袭与转移
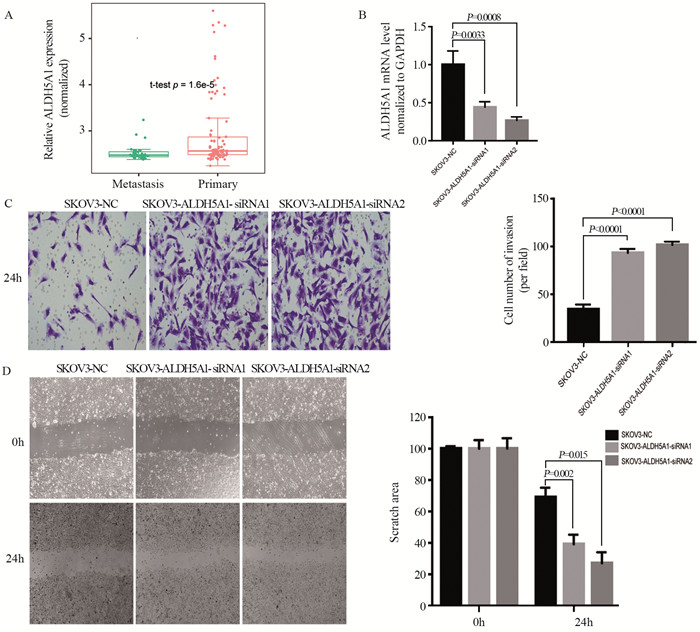
结果显示,转移灶组织(n=35)与原发灶(n=90)相比,转移灶的ALDH5A1表达水平显著下调(P=1.6×10-5),见图 1A。
![]() 图 1 ALDH5A1下调促进卵巢癌细胞侵袭与转移Figure 1 ALDH5A1 downregulation promoted metastasis and invasion of ovarian cancerA: transcription level of ALDH5A1 in the metastatic site of ovarian cancer (OC) (n=35) was markedly downregulated compared with that in the primary site (n=90), P=1.6×10-5; B: qRT-PCR analysis confirmed that ALDH5A1 expression was successfully downregulated in SKOV3-siRNA cells; C: Transwell assay showed that the invasion ability of OC cells dramatically increased; D: scratch-wound healing assay showed that the migratory ability of SKOV3 cells dramatically increased.
图 1 ALDH5A1下调促进卵巢癌细胞侵袭与转移Figure 1 ALDH5A1 downregulation promoted metastasis and invasion of ovarian cancerA: transcription level of ALDH5A1 in the metastatic site of ovarian cancer (OC) (n=35) was markedly downregulated compared with that in the primary site (n=90), P=1.6×10-5; B: qRT-PCR analysis confirmed that ALDH5A1 expression was successfully downregulated in SKOV3-siRNA cells; C: Transwell assay showed that the invasion ability of OC cells dramatically increased; D: scratch-wound healing assay showed that the migratory ability of SKOV3 cells dramatically increased.siRNA技术建立ALDH5A1表达下调的SKOV3卵巢癌细胞系,并用qRT-PCR验证:相比于SKOV3-NC组,SKOV3-siRNA1及SKOV3-siRNA2组中ALDH5A1mRNA表达水平下调,见图 1B。Transwell实验表明,随着ALDH5A1表达水平下调,卵巢癌细胞的迁移能力显著增强(P < 0.0001),见图 1C。划痕实验表明,卵巢癌细胞的迁移能力与ALDH5A1表达水平呈负相关(P < 0.01),见图 1D。
2.2 ALDH5A1与卵巢癌中ECM信号通路的关系
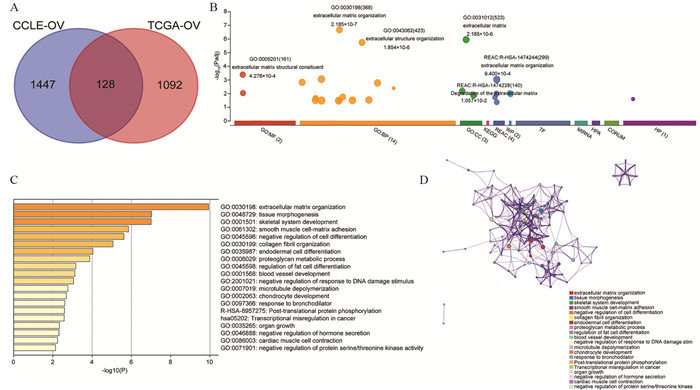
运用cBioPortal在线平台从CCLE数据库中提取了47个卵巢癌细胞株中1 575个ALDH5A1共表达基因,见附表 1,从TCGA数据库中的489个卵巢癌患者中提取了1 220个ALDH5A1共表达基因,见附表 2。通过取这两个共表达基因集的交集,发现有128个共同的共表达基因重叠,见图 2A、附表 3(附表 1~3请扫描本文OSID码)。对上述128个ALDH5A1的共表达基因进行功能富集分析,以识别共表达基因列表中可能存在的信号通路。首先,运用g: Profiler鉴定ALDH5A1与128个共表达基因的功能信息和富集途径及过程。如图 2B所示,生物进程(biological processes: BPs)方面,这些基因主要富集在ECM组织(GO: 0030198)和细胞外结构组织(GO: 0043062);细胞组成(cellular components: CCs)方面,这些基因主要富集在ECM通路中(GO: 0031012);分子功能(molecular functions: MFs)分析和Reactome(REAC)分析也显示这些基因在ECM结构成分(GO: 0005201)和ECM组织(REAC: R-HSA-1474244)中显著富集。同时用Metascape平台再次进行了GO分析。推测结果中与ALDH5A1关联密切的20种生命活动,其中富集最显著的基因集是ECM组织通路(GO: 0030198),见图 2C。Metascape绘制的共表达通路富集网路显示此20种生命活动彼此密切相关,见图 2D。
![]() 图 2 ALDH5A1基因功能富集分析以及其与卵巢癌中的共表达基因Figure 2 Gene-Ontology analysis of ALDH5A1 and coexpressed genes revealing the relationship between ALDH5A1 and ECM signaling pathways in OCA: Venn diagram showing the common coexpression genes that were found to overlap with ovarian cancer data derived from the CCLE database and TCGA database; B: g: Profiler was used to identify the functional information and enriched pathways and processes of ALDH5A1 and the 128 common co-expression genes; C: Metascape platform showing the top 20 putative biological processes; D: biological processes as revealed by Metascape in a network diagram.
图 2 ALDH5A1基因功能富集分析以及其与卵巢癌中的共表达基因Figure 2 Gene-Ontology analysis of ALDH5A1 and coexpressed genes revealing the relationship between ALDH5A1 and ECM signaling pathways in OCA: Venn diagram showing the common coexpression genes that were found to overlap with ovarian cancer data derived from the CCLE database and TCGA database; B: g: Profiler was used to identify the functional information and enriched pathways and processes of ALDH5A1 and the 128 common co-expression genes; C: Metascape platform showing the top 20 putative biological processes; D: biological processes as revealed by Metascape in a network diagram.2.3 卵巢癌患者TP53突变型别影响ALDH5A1与ECM途径中分子的相关性
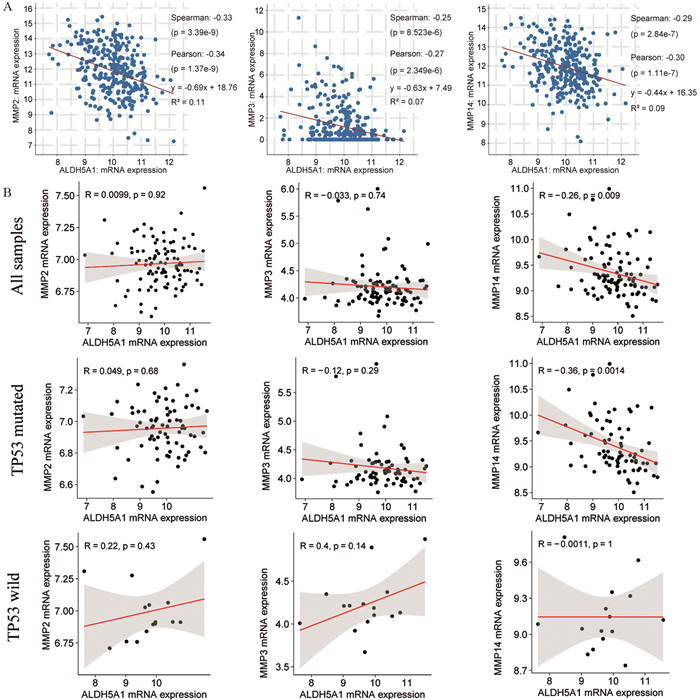
运用g: Profiler平台找到ECM通路中与ALDH5A1密切相关的基因MMP2、MMP3和MMP14。借助cBioportal平台中的卵巢癌数据库(TCGA, Firehouse Legacy数据集中卵巢浆液性囊腺癌临床资料)转录组数据“RNA Seq V2 RSEM” mRNA表达谱,对ALDH5A1与MMP2、MMP3、MMP14等基因的mRNA表达水平进行Sperman相关性分析。结果提示ALDH5A1与MMP2表达呈负相关(R=-0.34),ALDH5A1与MMP3表达呈负相关(R=-0.27),ALDH5A1与MMP14表达呈负相关(R=-0.30),见图 3A。通过分析GEO数据库中卵巢癌转录组芯片数据GSE63885,验证ALDH5A1与MMP2、MMP3、MMP14表达水平的相关性。发现ALDH5A1与MMPs表达水平的相关性与卵巢癌患者是否存在TP53突变有关:在所有卵巢癌患者(n=101)中,ALDH5A1仅与MMP14表达水平显著性负相关(R=-0.26, P=0.009);而存在TP53突变(n=75)的卵巢癌患者中,ALDH5A1与MMP14表达水平负相关更加显著(R=-0.36, P=0.0014);在TP53野生型(n=15)卵巢癌患者中,ALDH5A1与MMP2、MMP3、MMP14表达均无显著相关性,见图 3B。
![]() 图 3 ALDH5A1与ECM组织通路的共表达分析Figure 3 Coexpression analysis of ALDH5A1 and correlated genes among ECM pathwaysA: A negative correlation of mRNA expression was found between ALDH5A1 and MMP2, MMP3, MMP14. mRNA expression data was from "RNA Seq V2 RSEM" profile; B: Analysis of the GEO transcriptome chip data GSE63885 shows the correlation between ALDH5A1 and MMPs in relation to TP53 mutation type. The mRNA level is quantified by log2 (normalized counts + 1); P value was calculated by Person's correlation test.
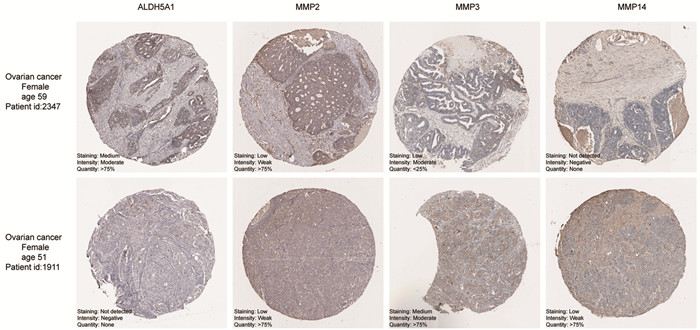
图 3 ALDH5A1与ECM组织通路的共表达分析Figure 3 Coexpression analysis of ALDH5A1 and correlated genes among ECM pathwaysA: A negative correlation of mRNA expression was found between ALDH5A1 and MMP2, MMP3, MMP14. mRNA expression data was from "RNA Seq V2 RSEM" profile; B: Analysis of the GEO transcriptome chip data GSE63885 shows the correlation between ALDH5A1 and MMPs in relation to TP53 mutation type. The mRNA level is quantified by log2 (normalized counts + 1); P value was calculated by Person's correlation test.一方面,我们从HPA数据库中获取卵巢癌TMA队列,临床样本免疫组织化学染色结果从蛋白水平验证卵巢癌患者ALDH5A1和MMPs表达水平呈负相关:ALDH5A1低表达组(Stain: Not detected/Intensity: Negative)中MMP14表达水平(Stain: low/Intensity: Weak)相较于高表达组(Stain: Medium/Intensity: Moderate)MMP14表达水平(Stain: Not detected/Intensity: Negative)有所提升,与转录组数据分析结果一致,见图 4。
2.4 ALDH5A1低表达的卵巢癌患者预后
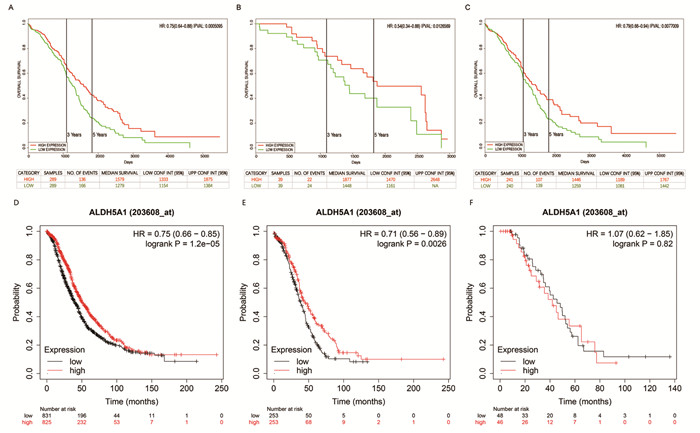
在卵巢癌患者中ALDH5A1表达较低者预后较差。ALDH5A1高水平卵巢癌患者的总生存率明显高于ALDH5A1低水平卵巢癌患者(HR=0.75(0.64~0.88), P=0.0005),见图 5A。病理Ⅱ级和Ⅲ级患者ALDH5A1高表达与OS改善相关(HR=0.54(0.34~0.88), P=0.0127; HR=0.79(0.66~0.94), P=0.0077),见图 5B~C。
![]() 图 5 ALDH5A1 mRNA表达水平与卵巢癌患者预后相关分析Figure 5 Relative analysis between ALDH5A1 mRNA expression and the prognosis of OC patientsA-C: data from TCGA database. A: all OC patients (n=578); B: gradeⅡ OC patients (n=78); C: gradeⅢ OC patients (n=481); D-F: data from Kaplan-Meier plotter online trancriptome chip data; D: all OC patients (n=1656); E: TP53-mutated OC patients (n=506); F: TP53-wild OC patients (n=94).
图 5 ALDH5A1 mRNA表达水平与卵巢癌患者预后相关分析Figure 5 Relative analysis between ALDH5A1 mRNA expression and the prognosis of OC patientsA-C: data from TCGA database. A: all OC patients (n=578); B: gradeⅡ OC patients (n=78); C: gradeⅢ OC patients (n=481); D-F: data from Kaplan-Meier plotter online trancriptome chip data; D: all OC patients (n=1656); E: TP53-mutated OC patients (n=506); F: TP53-wild OC patients (n=94).TP53突变型患者的ALDH5A1表达与总生存时间相关性更为显著(HR=0.71(0.56~0.89), P=0.0026),见图 5E,而TP53野生型卵巢癌患者的ALDH5A1表达则与总生存时间无显著相关(HR=1.07(0.62~1.85), P=0.82),见图 5F。
3 讨论
真核细胞的线粒体是一种重要的细胞器,在调节细胞增殖、分化、凋亡过程中发挥关键作用[6]。ALDH5A1属于ALDHs家族,编码产物为SSADH,后者对于线粒体代谢中GABA的降解起着非常重要的作用,功能缺陷或降低可导致GABA和GHB在体内蓄积。早在2001年即有研究报道,卵巢癌患者尿液中可观察到GABA浓度升高[14]。Hilvo等[6]发现高级别浆液性卵巢癌患者中GHB有异常蓄积现象,并证明这种蓄积现象是由ALDH5A1基因突变和活性降低引起的。尽管多项研究一致认为ALDH5A1与卵巢癌的发生发展相关,但卵巢癌中ALDH5A1作用的具体分子机制尚不清楚。
本研究发现与原发灶相比,卵巢癌患者转移组织中ALDH5A1的表达水平降低,提示细胞中ALDH5A1表达下调可能促进卵巢癌的侵袭和迁移。为深入了解ALDH5A1的功能,我们进行了功能富集分析,结果显示ALDH5A1共表达基因主要与ECM通路相关。ECM是由细胞合成并分泌到细胞外,分布在细胞表面或细胞之间的大分子物质,由基底膜(basement membrane, BM)和细胞间基质组成,是阻止肿瘤细胞转移的重要组织屏障。肿瘤细胞侵袭转移的首要条件是降解ECM和破坏BM,而MMPs是降解ECM最重要的蛋白酶类[15]。
近年来许多研究表明MMPs和肿瘤的发生发展密切相关,MMP2蛋白在卵巢癌转移中起早期应答蛋白的作用[16-18]。MMP14在基底膜和间质组织迁移过程中细胞外基质降解中发挥着核心作用[19],它刺激肿瘤-间质信号通路并促进卵巢癌细胞上的血管生成和肿瘤生长[20]。本研究从转录组表达数据、HPA免疫组织化学样本、生存分析数据发现,ALDH5A1下调促进卵巢癌细胞的侵袭和迁移与ECM通路中的关键基因MMP14相关,且这种影响可能与卵巢癌患者TP53突变有关。
综上所述,本研究发现ALDH5A1表达下调与卵巢癌转移密切相关,并初步探讨了ALDH5A1下调可能通过ECM通路影响卵巢癌细胞的侵袭转移能力,提示卵巢癌患者预后不良,具体分子机制尚需进一步研究。本研究提示ALDH5A1可能作为卵巢癌转移标志物以及潜在的治疗靶点。
Competing interests: The authors declare that they have no competing interests.作者贡献:赵桐:研究设计、实验实施和论文撰写田训:研究思路指导和论文审较曹晨:研究设计、实验指导和论文审校 -
-
[1] Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71(3): 209-249. doi: 10.3322/caac.21660
[2] Torre LA, Trabert B, Desantis CE, et al. Ovarian cancer statistics[J]. CA Cancer J Clin, 2018, 68(4): 284-296. doi: 10.3322/caac.21456
[3] Yang Y, Karakhanova S, Hartwig W, et al. Mitochondria and Mitochondrial ROS in Cancer: Novel Targets for Anticancer Therapy[J]. J Cell Physiol, 2016, 231(12): 2570-2581. doi: 10.1002/jcp.25349
[4] Zhou Z, Ibekwe E, Chornenkyy Y. Metabolic Alterations in Cancer Cells and the Emerging Role of Oncometabolites as Drivers of Neoplastic Change[J]. Antioxidants (Basel), 2018, 7(1): 16. doi: 10.3390/antiox7010016
[5] Egan G, Khan DH, Lee JB, et al. Mitochondrial and Metabolic Pathways Regulate Nuclear Gene Expression to Control Differentiation, Stem Cell Function, and Immune Response in Leukemia[J]. Cancer Discov, 2021, 11(5): 1052-1066. doi: 10.1158/2159-8290.CD-20-1227
[6] Hilvo M, De Santiago I, Gopalacharyulu P, et al. Accumulated Metabolites of Hydroxybutyric Acid Serve as Diagnostic and Prognostic Biomarkers of Ovarian High-Grade Serous Carcinomas[J]. Cancer Res, 2016, 76(4): 796-804. doi: 10.1158/0008-5472.CAN-15-2298
[7] Deng XY, Gan XX, Feng JH, et al. ALDH5A1 acts as a tumour promoter and has a prognostic impact in papillary thyroid carcinoma[J]. Cell Biochem Funct, 2021, 39(2): 317-325. doi: 10.1002/cbf.3584
[8] Kaur H, Mao S, Li Q, et al. RNA-Seq of human breast ductal carcinoma in situ models reveals aldehyde dehydrogenase isoform 5A1 as a novel potential target[J]. PLoS One, 2012, 7(12): e50249. doi: 10.1371/journal.pone.0050249
[9] Tian X, Han Y, Yu L, et al. Decreased expression of ALDH5A1 predicts prognosis in patients with ovarian cancer[J]. Cancer Biol Ther, 2017, 18(4): 245-251. doi: 10.1080/15384047.2017.1295175
[10] Goswami CP, Nakshatri H. PROGgeneV2: enhancements on the existing database[J]. BMC Cancer, 2014, 14: 970. doi: 10.1186/1471-2407-14-970
[11] Cerami E, Gao J, Dogrusoz U, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data[J]. Cancer Discov, 2012, 2(5): 401-404. doi: 10.1158/2159-8290.CD-12-0095
[12] Raudvere U, Kolberg L, Kuzmin I, et al. g: Profiler: a web server for functional enrichment analysis and conversions of gene lists (2019 update)[J]. Nucleic Acids Res, 2019, 47(W1): W191-W198. doi: 10.1093/nar/gkz369
[13] Zhou Y, Zhou B, Pache L, et al. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets[J]. Nat Commun, 2019, 10(1): 1523. doi: 10.1038/s41467-019-09234-6
[14] Nicholson-Guthrie CS, Guthrie GD, Sutton GP, et al. Urine GABA levels in ovarian cancer patients: elevated GABA in malignancy[J]. Cancer Lett, 2001, 162(1): 27-30. doi: 10.1016/S0304-3835(00)00620-0
[15] 胡梅艳, 孙晓红. 细胞外基质、基质金属蛋白酶与恶性肿瘤关系的研究进展[J]. 肿瘤药学, 2016, 6(1): 26-30. https://www.cnki.com.cn/Article/CJFDTOTAL-LIYX201601008.htm Hu YM, Sun XH. The research progress of relationship between ECM, MMPs and malignant tumor[J]. Zhong Liu Yao Xue, 2016, 6(1): 26-30. https://www.cnki.com.cn/Article/CJFDTOTAL-LIYX201601008.htm
[16] Wang X, Yang B, She Y, et al. The lncRNA TP73-AS1 promotes ovarian cancer cell proliferation and metastasis via modulation of MMP2 and MMP9[J]. J Cell Biochem, 2018, 119(9): 7790-7799. doi: 10.1002/jcb.27158
[17] Gonzalez-Villasana V, Fuentes-Mattei E, Ivan C, et al. Rac1/Pak1/p38/MMP-2 Axis Regulates Angiogenesis in Ovarian Cancer[J]. Clin Cancer Res, 2015, 21(9): 2127-2137. doi: 10.1158/1078-0432.CCR-14-2279
[18] Qiu J, Ye L, Ding J, et al. Effects of oestrogen on long noncoding RNA expression in oestrogen receptor alpha-positive ovarian cancer cells[J]. J Steroid Biochem Mol Biol, 2014, 141: 60-70. doi: 10.1016/j.jsbmb.2013.12.017
[19] Castro-Castro A, Marchesin V, Monteiro P, et al. Cellular and Molecular Mechanisms of MT1-MMP-Dependent Cancer Cell Invasion[J]. Annu Rev Cell Dev Biol, 2016, 32: 555-576. doi: 10.1146/annurev-cellbio-111315-125227
[20] Kaimal R, Aljumaily R, Tressel SL, et al. Selective blockade of matrix metalloprotease-14 with a monoclonal antibody abrogates invasion, angiogenesis, and tumor growth in ovarian cancer[J]. Cancer Res, 2013, 73(8): 2457-2467. doi: 10.1158/0008-5472.CAN-12-1426



 下载:
下载: